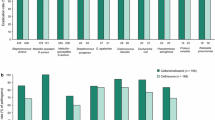
Summary
Five open-label and three double-blind studies were conducted to evaluate the effectiveness of cefadroxil (Duricef®) in treatment of infections of the skin and skin structures. After a ten-day treatment period at dosages of 1,000 mg qd and 500 mg or 1,000 mg bid, favorable clinical response to treatment was noted in 99% of the patients treated. A satisfactory bacteriologic response was observed in 96%. Efficacy did not vary with the treatment regimen. Results obtained with a once- or twice-daily dosage regimen of cefadroxil were equivalent to those achieved with a four-times-daily regimen of cephalexin. These studies provide overall evidence of the efficacy of cefadroxil in the treatment of skin and skin structure infections. Particularly, its effectiveness when given only once daily represents an important advance in antimicrobial therapy.
Zusammenfassung
Um die Wirksamkeit von Cefadroxil (Duricef®) in der Behandlung von Infektionen der Haut und ihrer Anhangsgebilde zu untersuchen, wurden fünf offene und drei Doppelblindstudien durchgeführt. Nach einer zehntägigen Behandlungszeit mit Dosen von 1000 mg einmal täglich und 500 oder 1000 mg zweimal täglich wurde bei 99% der behandelten Patienten ein günstiges klinisches Ansprechen auf die Therapie beobachtet. Die Behandlungsschemata unterschieden sich nicht in ihrer Effektivität. Die mit einem ein- oder zweimal täglichen Dosierungsmodus erreichten therapeutischen Ergebnisse waren ebenso gut wie die eines viermal täglichen Dosisschemas von Cephalexin. In den vorliegenden Untersuchungen wird die Wirksamkeit von Cefadroxil bei Infektionen der Haut unter Beweis gestellt. Die Wirksamkeit der Substanz bei einer einzigen täglichen Dosis stellt einen bedeutsamen Fortschritt in der antimikrobiellen Therapie dar.
Similar content being viewed by others
Literature
Mead Johnson Pharmaceutical Division: Cefadroxil (Duricef®) Prescribing Information.
Buck, R. E., Price, K. E. Cefadroxil, a new broad-spectrum cephalosporin. Antimicrob. Agents Chemother. 11 (1977) 324–330.
Gordon, W. E. Efficacy and safety of cefadroxil. J. Ken. Med. Ass. 76 (1978) 121–123.
Bolding, O. T. Clinical comparison of cefadroxil, a new oral cephalosporin, and cephalexin in uncomplicated urinary tract infection. Urology 12 (1979) 321–324.
Weingarten, C. M. Double-blind study of cefadroxil in uncomplicated urinary tract infection. J. Cont. Educ. Fam. Med. 25 (1979) 51–53.
Unpublished data for studies of serum and skin blister levels of cefadroxil. Mead Johnson Pharmaceutical Division Report. July 10, 1978.
Santella, P. J., Berman, E. Cefadroxil: Sustained antimicrobial effect in bacterial infections. A review of clinical studies. Curr. Ther. Res. 23 (1978) 148–158.
Bauer, A. W., Kirby, W. M., Sherris, J. C., et al. Antibiotic susceptibility testing by a standard single disc method. Am. J. Clin. Path. 45 (1965) 493–496.
Pfeffer, M., Jackson, A., Ximenes, J., et al. Comparative human oral clinical pharmacology of cefadroxil, cephalexin, and cephradine. Antimicrob. Agents Chemother. 11 (1977) 331–338.
Richards, D., Jr.: Bioavailability and excretion characteristics of cefadroxil. Postgrad. med. Comm. (1979) 27–30.
Hausman, M. S.: Evaluation of cefadroxil administered in a high-compliance dose regimen. Postgrad. med. Comm. (1979) 31–35.
Author information
Authors and Affiliations
Additional information
Erstveröffentlichung: Postgrad. Med. Communications, Rationale and Clinical Experience (1979) 41.
Rights and permissions
About this article
Cite this article
Henness, D.M., Gordon, W.E. Effective once- or twice-daily treatment of skin and skin structure infections with a new cephalosporin (cefadroxil). Infection 8 (Suppl 5), S633–S637 (1980). https://doi.org/10.1007/BF01639686
Issue Date:
DOI: https://doi.org/10.1007/BF01639686