Summary
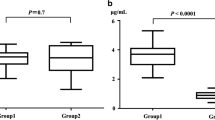
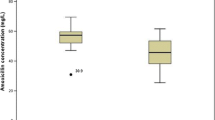
The recommended neonatal dosage regime for amikacin of 15 mg/kg/day was found to be unsatisfactory in four neonates and a loading dose of 10 mg/kg did not improve levels in two further newborns with normal renal function. Fourteen neonates on 20 mg/kg/day regime achieved satisfactory levels; peak values were 21.5±4.5 µg/ml and trough levels 3.34±2.05 µg/ml. Peak time ranged from 17 to 60 minutes and varied with the dose given. The half-life was related to plasma creatinine, urea, post-natal age and weight. Thirteen neonates with pre-existing renal impairment were managed by alteration of dosage interval or quantity given. Side-effects were confined to eosinophilia in six infants, and no definite nephrotoxicity or gross eighth nerve damage was demonstrated.
Zusammenfassung
Es zeigte sich, daß das für Neugeborene empfohlene Dosierungsschema für Amikacin von 15 mg/kg/Tag bei vier Neugeborenen nicht zufriedenstellend war und eine Anfangsdosis von 10 mg/kg bei zwei weiteren Neugeborenen mit normaler Nierenfunktion nicht zu einer Verbesserung der Spiegel führte. Vierzehn Neugeborene erreichten mit 20 mg/kg/Tag zufriedenstellende Spiegel; die Spitzenwerte betrugen 21,5±4,5 µg/ml und die mittleren Spiegel lagen bei 3,34±2,05 µg/ml. Die Maxima wurden zwischen 17 und 60 Minuten erreicht und wechselten mit der verabreichten Dosis. Die Halbwertszeit stand in Relation zu Plasmacreatinin, Harnstoff, Lebensalter und Gewicht. Dreizehn Neugeborene mit vorbestehender Nierenstörung konnten wir durch Änderung des Dosierungsintervalls oder der verabreichten Dosis behandeln. Nebenwirkungen beschränkten sich auf Eosinophilie bei sechs Kindern. Zeichen für eine ausgesprochene Nephrotoxizität oder erhebliche Störungen des achten Hirnnerven konnten nicht festgestellt werden.
Similar content being viewed by others
Literature
McCracken, G. H., Howard, J. B. Pharmacological evaluation of amikacin in neonates. Antimicrob. Agents Chemother. 8 (1975) 86–90.
Sardemann, H. H., Colding, H., Herdel, J., Kampmann, J. P., Hvidberg, E. F., Vejlsgaard, R. Kinetics and dose calculations of amikacin in the newborn. Clin. Pharmacol. Ther. 20 (1976) 59–66.
Chiu, T. W., Pfeffer, M., Badri, M. S., Gamson, D., Ayoub, E. M.: Single dose pharmacokinetics evaluation of amikacin in neonates. Am. J. Med., U.S. Amikacin Symposium (1977) 84–89.
Want, S. V., Jones, R. A. K., Darrell, J. H. Amikacin dosage in the preterm newborn. J. Antimicrob. Chemother. 5 (1979) 527–530.
Dubowitz, L., Dubowitz, V.: Clinical assessment of gestational age in the newborn infant. J. Pediatr. 77 (1970).
Haas, M. J., Davies, J. Enzymatic acetylation as a means of determining serum aminoglycoside determinations. Antimicrob. Agents Chemother. 4 (1973) 497–499.
Metis, S. (ed.): Paediatric Clinical Chemistry. Ross Laboratories New York, 1977, pp. 89–91.
Nie, N. H., Hall, C. H., Jenkins, J. C., Steinbrenner, K., Bert, D. H. S.P.S.S. 2nd edition, 1970, McGraw-Hill, New York.
Trujillo, H.: Amikacin in paediatric infections. Current Chemotherapy, Proceedings of the 10th International Congress of Chemotherapy (1977) 1000–1002.
Smith, C. R., Baughman, K. L., Edwards, C. Q., Rogers, J. F., Leitman, P. S. Controlled comparison of amikacin and gentamicin. N. Engl. J. Med. 296 (1977) 349–353.
Dittert, L. W.: Pharmacokinetics of aminoglycosides: General considerations. Am. J. Med., U.S. Amikacin Symposium (1976) 77–83.
Pierce, A. K., Sanford, J. P. Anaerobic gram-negative bacillary pneumonias. Am. Rev. Respir. Dis. 110 (1974) 647–650.
Noone, P., Parsons, T. M. C. Experience in monitoring gentamicin therapy during treatment of serious gram-negative sepsis. Br. Med. J. 1 (1974) 477–484.
Schaffer, A., Avery, M. Diseases of the newborn. W. B. Saunders & Co., Philadelphia, 1977, pp. 764–771.
Fie, P. K., Neu, H. C.: Amikacin in vitro activity against multiresistant bacteria used singly and in combination with penicillins. Am. J. Med., U.S. Amikacin Symposium (1976) 46–53.
Drasar, F. A., Farrell, W., Maskell, J., Williams, J. D. Tobramycin, amikacin, sisomicin and gentamicin-resistant gram-negative rods. Br. Med. J. 2 (1976) 1284–1287.
Clark, J. T., Libke, R. D., Regamy, C., Kirby, W. M. Comparative pharmacokinetics of amikacin and kanamycin. Clin. Pharmacol. Ther. 15 (1974) 610–616.
Myers, M. G., Roberts, R. J., Mirhij, N. J. Effect of gestational age, birth weight and hypoxia on pharmacokinetics of amikacin in serum of infants. Antimicrob. Agents Chemother. 11 (1977) 1027–1032.
Bodey, G. P., Stewart, D. In vitro studies of BBK8, a new aminoglycoside antibiotic. Antimicrob. Agents Chemother. 4 (1973) 186–190.
McCracken, G. H., Nelson, J. D. Antimicrobial therapy for new-borns. London, Grune and Stratton, 1977, p. 44.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cookson, B., Williams, J.D., Tripp, J. et al. Evaluation of amikacin dosage regimes in the low and very low birthweight newborn. Infection 8 (Suppl 3), S239–S242 (1980). https://doi.org/10.1007/BF01639588
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01639588