Summary
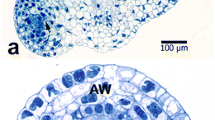
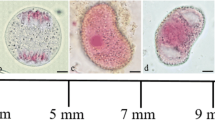
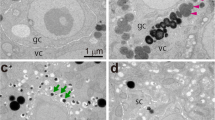
Using a combination of electron microscopic and cytochemical techniques, the activity of the lytic enzyme acid phosphatase during male meiosis and sporogenesis inDatura innoxia Mill, has been followed. Evidence for digestion of cytoplasmic structures such as ribosomes and membranes during meiotic prophase and in the vacuolate microspores are presented. Before the onset of meiosis, acid phosphatase activity was observed chiefly in the walls of the pollen mother cell. During meiosis, due to the presence of a thick callose wall around the meiocytes, the enzyme activity could not be detected although the presence of numerous “autophagic type vacuoles” suggest lytic activity within these vacuoles. A positive reaction was, however, found in the adjacent somatic tissues. Also, low activity was observed in the small vacuoles of the young microspore, and the reaction was very intense in the large central vacuole of the old microspore which has a peripheral nucleus. In the young bicellular or mature pollen the activity was mainly observed in the small vacuoles. Other organelles showed no reaction. The significance of this cellular autophagy which is only observed at the meiotic prophase and at the vacuolate stage of the microspore is discussed, particularly in relation to the sporophyte-gametophyte transition.
Similar content being viewed by others
Abbreviations
- PMC:
-
pollen mother cell
- TEM:
-
transmission electron microscope
References
Buvat R, Robert G (1979) Vacuoles formation in the actively growing root meristem of barley (Hordeum sativum). Am J Bot 66: 1219–1237
Cronshaw J, Bentwood BJ (1977) Cytochemical localization of ßglycerophosphatase in the xylem ofNicotiana tabacum andPisum sativum. Biol Cell 28: 65–74
Dickinson HG (1987) The physiology and biochemistry of meiosis in the anther. Int Rev Cytol. 107: 79–108
Esau K, Charvat ID (1975) An ultrastructural study of acid phosphatase localization in cells ofPhaseolus vulgaris phloem by the use of the azo dye method. Tissue Cell 7: 619–630
Gahan PB (1965) Histochemical evidence for the presence of lysosome like particles in root meristem cells ofVicia faba. J Exp Bot 16: 350–355
Gartner PJ, Nagl W (1980) Acid phosphatase activity in plastides (Plastolysomes) of senescing embryo-suspensor cells. Planta 149: 341–349
Gomori G (1950) An improved histochemical technique for acid phosphatase. Stain Technol 25: 81–85
Hall LJ, Butt US (1968) Localization and kinetic properties of ßglycerophosphatase in barley roots. J Exp Bot 19: 276–287
Halperin W (1969) Ultrastructural localization of acid phosphatase in cultured ofDaucus carota. Planta 88: 91–102
Herman EM, Baumgartner B, Chrispels MJ (1981) Uptake and apparent digestion of cytoplasmic organelles by protein bodies (protein storage vacuoles) in mung bean cotyledons. Eur J Cell Biol 24: 226–235
Heslop-Harrison J (1972) Sexuality of angiosperms. In: Steward FC (ed) Plant physiology. A treatise, vol 6. Academic Press, New York, pp 139–289
— (1977) Ribosomes, membranes and organelles during meiosis in angiosperms. Philos Trans R Soc Lond [Biol] 277: 327–342
Holcomb GE, Hildebrandt AC, Evert RF (1967) Staining and acid phosphatase reactions of spherosomes in plant tissue culture cells. Am J Bot 54: 1204–1209
Horvat F (1969) La paroi de la cellule générative du grain de pollen. Pollen Spores 11: 181–201
Knox RB, Dickinson HG, Heslop-Harrison J (1970) Cytochemical observations on changes in RNA content and acid phosphatase activity during the meiotic prophase in the anther ofCosmos bipinnatus cav. Acta Bot Neerl 19: 1–6
Linskens HF (1966) Die Änderung des Proteins- und Enzym-Musters während der Pollenmeiose und Pollenentwicklung. Physiologische Untersuchungen zur Reifeteilung. Planta 69: 79–91
Lugon M, Goldberg R, Guern J (1974) Les fragments de paroi de cellules d'Acerpseudoplatanus, matériel d'étude de l'activite des phosphatases pariétales. CR Hedb Séance Acad Sci 278: 1221–1224
Mackenzie A, Heslop-Harrison J, Dickinson HG (1967) Elimination of ribosomes during meiotic prophase. Nature 215: 997–999
Marty F (1982) Lysosomes: le modéle vacuolaire des plantes. Bull Soc Bot Fr 129, Actual Bot 3: 35–54
—, Branton D, Leigh RA (1980) Plant vacuoles. In: Stumpf PK, Conn EE (eds) The biochemistry of plants, vol 1. Academic Press, New York, pp 625–658
Mathivet V (1984) Aspects biochimiques et cytochimiques du pollen deDatura et du Tabac, au cours du développement gamétophytique et androgénétique. DEA Univ Paris VI, pp 1–75
Matile P (1975) Biochemistry and function of vacuoles. Ann Rev Plant Physiol 29: 193–213
—, Spichiger J (1968) Lysosomal enzymes in spherosomes (oil droplets) of tabacco endosperm. Z Pflanzenphysiol 58: 277–280
Nagl W (1977) Plastolysomes-plastids involved in the autolysis of the embryo-suspensor inPhaseolus. Z Pflanzenphysiol 85: 45–51
Sangwan RS (1986) Formation and cytochemistry of nuclear vacuoles during meiosis inDatura. Eur J Cell Biol 40: 210–218
—, Camefort H (1982) Microsporogenesis inDatura metel L. Rev Cytol Biol Végét Bot 5: 265–282
—, Sangwan-Norreel BS (1987 a) Ultrastructural cytology of plastids in pollen grains of certain androgenic and nonandrogenic plants. Protoplasma 138: 11–22
— — (1987 b) Biochemical cytology of pollen embryogenesis. Int Rev Cytol 107: 221–272
Suzuki T, Sato S (1973) Properties of acid phosphatase in the cell wall of tobacco cells culturedin vitro. Plant Cell Physiol 14: 589–596
Van der Wilden W, Herman EM, Chrispeels MJ (1980) Protein bodies of mungbean cotyledons as autophagic organelles. Proc Natl Acad Sci USA 77: 428–433
Vithanaga HIMV, Knox RB (1976) Pollen-wall proteins: quantitative cytochemistry of the origins of intine and exine enzymes inBrassica oleracea. J Cell Sci 21: 423–435
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sangwan, R.S., Mathivet, V. & Vasseur, G. Ultrastructural localization of acid phosphatase during male meiosis and sporogenesis inDatura: Evidence for digestion of cytoplasmic structures in the vacuoles. Protoplasma 149, 38–46 (1989). https://doi.org/10.1007/BF01623981
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01623981