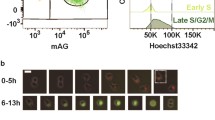
Summary
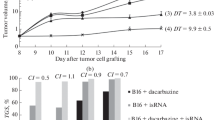
The responses of B16 melanoma in C57BL mice to cytotoxic agents, cyclophosphamide, doxorubicin, and dactinomycin, were studied by different methods both in vivo and in vitro. Either treated tumors were allowed to grow in vivo or cells were isolated and cultured in vitro afterwards. The tumor cells were also treated in vitro and assayed in vitro. For cyclophosphamide, a fairly good correlation was found among the dose responses measured: the tumor growth delay, incidence of giant cells, decrease in mitotic index, clonogenic cell survival of tumor cells treated in vivo and the survival of cells treated in vitro. For doxorubicin and dactinomycin, on the other hand, the tumor growth delay was not marked, the appearance of giant cells and decrease in mitotic index were minimal, and almost no decrease was found in the clonogenic cell survival for tumor treated in vivo, although the cells responded well to in vitro exposure. The results indicate substantial differences in the expression of damage according to the conditions of tumor cell growth and assay after treatment with different agents.
Similar content being viewed by others
References
Balconi G, Bossi A, Donelli MG, Filippeschi S, Franchi G, Morasca L, Garattini S (1973) Chemotherapy of a spontaneous mammary carcinoma in mice: relation between in vitro-in vivo activity and blood and tumor concentrations of several antitumor drugs. Cancer Chemother Rep 57:115–124
Begg AC (1980) Analysis of growth delay data: potential pitfalls. Br J Cancer 41, suppl IV:93–97
Begg AC, Smith KA (1984) A bioassay for cyclophosphamide in blood, lung and tumor. Br J Cancer 49:49–55
Begg AC, Fu KK, Kane LJ, Phillips TL (1980) Single-agent chemotherapy of a solid murine tumor assayed by growth delay and cell survival. Cancer Res 40:145–154
Courtenay VD, Mills J (1978) An in vitro colony assay for human tumors grown in immune-suppressed mice and treated with cytotoxic agents. Br J Cancer 37:261–268
Domeyer BE, Sladek NE (1980) Kinetics of cyclophosphamide bio-transformation in vivo. Cancer Res 40:174–180
Erlichman C, Vidgen D (1984) Cytotoxicity of adriamycin in MGH-U1 cells grown as monolayer cultures, spheroids, and xenografts in immune-deprived mice. Cancer Res 44:5369–5375
Freireich EJ, Grehan EA, Rall DP, Schmidt LH, Skipper HE (1966) Quantitative comparison of toxicity of anticancer agents in mouse, rat, hamster, dog, monkey, and man. Cancer Chemother Rep 50:219–245
Furuse T, Kasuga T (1983) Difference in3H-thymidine incorporation after irradiation between murine B16 melanoma and squamous cell carcinoma in vivo. Gann 74:232–239
Kura S, Sasaki H, Aramaki R, Yoshinaga H (1978) Binucleated cell formation induced by X-irradiation. Radiat Res 76:83–94
Li X, Paulus G, Atassi G, Buyssens N (1984) Growth response of B16 melanoma to in vivo treatment with 1-(2-chloroethyl)-3-cyclohexyl-1-nitrosourea (CCNU) at the initial stage after tumor transplantation. Am J Pathol 115:403–411
Martin WMC, Macnally NJ (1980) Cytotoxicity of adriamycin to tumor cells in vivo and in vitro. Br J Cancer 42:881–889
Piro AJ, Taylor CC, Belli JA (1975) Interaction between radiation and drug damage in mammalian cells I. delayed expression of actinomycin D/X-ray effects in exponential and plateau phase cells. Radiat Res 63:346–362
Puck TT, Marcus PI (1956) Action of X-rays on mammalian cells. J Exp Med 103:653–666
Rockwell S (1980) In vivo-in vitro tumor cell lines: characteristics and limitations as models for human cancer. Br J Cancer 41, suppl IV:118–122
Rockwell S, Kallman RF (1973) Cellular radiosensitivity and tumor radiation response in the EMT6 tumor cell system. Radiat Res 53:281–294
Roper PR, Drewinko B (1976) Comparison of in vitro methods to determine drug-induced cell lethality. Cancer Res 36:2182–2188
Schwartz HS, Sodergren JE, Ambaye RY (1968) Actinomycin D: drug concentrations and actions in mouse tissues and tumors. Cancer Res 28:192–197
Sharkis SJ, Santos GW, Colvin M (1980) Elimination of acute myelogenous leukemic cells from marrow and tumor suspensions in the rat with 4-hydroperoxycyclophosphamide. Blood 55:521–523
Shimoyama M, Niitani H, Kimura K (1975) Chemotherapeutic effect of cyclophosphamide and its related active compounds 2. studies on cell-kill kinetics and chemotherapeutic effects of 4-hydroperoxy cyclophosphamide and 4-hydroperoxy isophosphamide (in Japanese). Gan To Kagakuryouhou 2:883–888
Steel GG, Courtenay VD, Peckham MJ (1983) The response to chemotherapy of a variety of human tumor xenografts. Br J Cancer 47:1–13
Stephens TC, Peacock JH, Steel GG (1977) Cell survival in B16 melanoma after treatment with combination of cytotoxic agents: lack of potentiation. Br J Cancer 36:84–93
Thomlinson RH, Craddock EA (1967) The gross response of experimental tumor to single doses of X-rays. Br J Cancer 21:108–123
Twentyman PR (1977) Sensitivity to cytotoxic agents of the EMT6 tumor in vivo: tumor volume versus in vitro plating. 1. cyclophosphamide. Br J Cancer 35:208–217
Twentyman PR (1979) Timing of assays: an important consideration in the determination of clonogenic cell survival both in vitro and in vivo. Int J Radiat Oncol Biol Phys 5:1213–1220
Twentyman PR (1980) Experimental chemotherapy studies: intercomparison of assays. Br J Cancer 41, suppl IV:279–287
Von Hoff DD, Casper J, Bradley E, Sandbach J, Jones D, Makuch R (1981) Association between human tumor colony-forming assay results and response of an individual patient's tumor to chemotherapy. Am J Med 70:1027–1032
Wassermann K, Rasmussen SN (1987) Comparative responsiveness and pharmacokinetics of doxorubicin in human tumor xenografts. Eur J Cancer Clin Oncol 23:303–309
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kuwashima, Y., Matsubara, O. & Kasuga, T. Responses of a murine B16 melanoma to pharmacotherapy studied and compared with different assay systems. J Cancer Res Clin Oncol 116, 173–178 (1990). https://doi.org/10.1007/BF01612673
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01612673