Summary
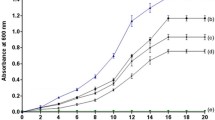
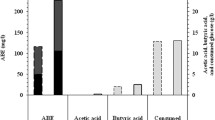
A total of 1229 cultures, including 230 actinomycetes, 508 other bacteria, 12 fungi and 479 yeasts were screened for their ability to oxidize the isopropyl side chain of 2-phenyl propane (cumene). Four strains of actinomycetes and six strains of bacteria but no yeasts were found positive in converting 2-phenyl propane to its oxygenated products. Eight strains oxidized cumene through the alkyl side chain producing 2-phenyl-1-propanol. TwoBacillus strains oxidized cumene to an oxygenated product.Pseudomonas oleovorans NRRL B-3429 exhibited the highest alkyl side chain oxidation activity. The optimum reaction conditions for strain B-3429 are: 25 °C, pH 6.5 and 48 h of reaction. Octane-grown cells of strain B-3429 produced higher product yields (about 7.2-fold) than the glucose-grown cells. Prolonged incubation resulted in an increase in 2-phenyl-1-propionic acid production at the expense of 2-phenyl-1-propanol. The yield of 2-phenyl-1-propanol plus 2-phenyl-1-propionic acid was 5.1%. Reaction in the presence of methanol favored the accumulation of 2-phenyl-1-propionic acid and also increased the total yield. (The yield of 2-phenyl-1-propanol plus 2-phenyl-1-propionic acid was 14.9%.) Structures of the reaction products were confirmed by GC/MS and GC/IR analyses. Products contained 92% R(−) isomer.
Similar content being viewed by others
References
Abbott, B.J. and C.T. Hou. 1973. Oxidation of 1-alkenes to 1,2-epoxyalkanes byPseudomonas oleovorans. Appl. Microbiol. 26: 86–91.
Barton, M.J., J.P. Hamman, K.C. Fichter and G.J. Calton. 1990. Enzymic resolution of R,S-2-(4-hydroxyphenoxy) propionic acid. Enzyme Microbiol. Technol. 12: 577–583.
Davis, J.B. and R.L. Raymond. 1961. Oxidation of alkylsubstituted cyclic hydrocarbons by aNocardia during growth on n-alkanes. Appl. Microbiol. 9: 383–388.
Gibson, D.T., B. Gschwendt, W.K. Yeh and V.M. Kobal. 1973. Initial reactions in the oxidation of ethylbenzene byPseudomonas putida. Biochemistry 12: 1520–1528.
Hou, C.T. and T.M. Johnston. 1992. Screening of lipase activities with cultures from the ARS Culture Collection. J. Am. Oil Chem. Soc. 69: 1088–1097.
Hou, C.T., R.N. Patel, A.I. Laskin and N. Barnabe. 1982. Epoxidation of alkenes by methane monoxygenase: generation and regeneration of cofactor, NADH, by dehydrogenases. J. Appl. Biochem. 4: 379–383.
Hou, C.T., R.N. Patel, A.I. Laskin, I. Marczak and N. Barnabe. 1982. Epoxidation and hydroxylation of C4 and C5 branch-chain alkenes and alkanes by methylotrophic bacteria. Dev. Ind. Microbiol. 23: 477–482.
Jigami, Y., Y. Kawasaki, T. Omori and Y. Minoda. 1979. Coexistence of different pathways in the metabolism of n-propylbenzene byPseudomonas sp. Appl. Environ. Microbiol. 38: 783–788.
Jigami, Y., T. Omori and Y. Minoda. 1975. The degradation of isopropylbenzene and isobutylbenzene byPseudomonas sp. Agric. Biol. Chem. 39: 1781–1788.
Klifford, K.H., G.T. Phillips and A.F. Marx. 1989. Process for the preparation of substituted phenoxy propanoic acid. Eur. Patent Public. 0319100A2 (07.06.89).
Kuge, Y., K. Mochida and T. Uwajima. 1991. Microbial hydroxylation of 2-phenylpropionic acid. Agric. Biol. Chem. 55: 1099–1104.
Sih, C.J., Q.-M. Gu, G. Fulling, S.-H. Wu and D.R. Reddy. 1988. The use of microbial enzymes for the synthesis of optically active pharmaceuticals. Dev. Ind. Microbiol. 29: 221–229.
Webley, D.M., R.B. Duff and V.C. Farmer. 1956. Evidence for oxidation in the metabolism of saturated aliphatic hydrocarbons by soil species ofNocardia. Nature (London) 178: 1467.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hou, C.T., Seymour, T.A. & Bagby, M.O. Microbial oxidation of cumene. Journal of Industrial Microbiology 13, 97–102 (1994). https://doi.org/10.1007/BF01584105
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01584105