Abstract
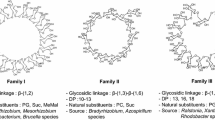
We have previously shown that species ofBradyrhizobium synthesize a novel class of cyclic beta glucans which contains both beta-1,6 and beta-1,3 glycosidic linkages [Miller KJ, Gore RS, Johnson R, Benesi AJ, Reinhold VN (1990) J Bacteriol 172:136–142]. In the present study we show that these cell-associated glucans are localized within the periplasmic compartment and that the biosynthesis of these glucans is osmotically regulated.
Similar content being viewed by others
Literature Cited
Abe M, Amemura A, Higashi S (1982) Studies on cyclic beta-1,2-glucan obtained from periplasmic space ofRhizobium trifolli cells. Plant Soil 64:315–324
Ames GF-L, Prody C, Kustu S (1984) Simple, rapid, and quantitative release of periplasmic proteins by chloroform. J Bacteriol 160:1181–1183
Batley M, Redmond JW, Djordjevic SP, Rolfe BG (1987) Characterization of glycerophosphorylated cyclic beta-1,2-glucans from a fast-growingRhizobium species. Biochim Biophys Acta 901:119–126
Cangelosi GA, Martinetti G, Nester EW (1990) Osmosensitivity phenotypes ofAgrobacterium tumefaciens mutants that lack periplasmic beta-1,2-glucan. J Bacteriol 172:2172–2174
Douglas CJ, Staneloni RJ, Rubin RA, Nester EW (1985) Identification and genetic analysis of anAgrobacterium tumefaciens chromosomal virulence region. J Bacteriol 161:850–860
Dylan T, Ielpi L, Stanfield S, Kashyap L, Douglas C, Yanofsky M, Nester E, Helinski DR, Ditta G (1986)Rhizobium meliloti genes required for nodule development are related to chromosomal virulence genes inAgrobacterium tumefaciens, Proc Natl Acad Sci USA 83:4403–4407
Dylan T, Helinski DP, Ditta GS (1990) Hypoosmotic adaptation inRhizobium meliloti requires beta-(1–2)-glucan. J Bacteriol 172:1400–1408
Dylan T, Nagpal P, Helinski DR, Ditta GS (1990) Symbiotic pseudorevertants ofRhizobium meliloti ndv mutants. J Bacteriol 172:1409–1417
Hanson RS, Phillips JA (1981) Chemical composition, In: Gerhardt P, Murray RGE, Costilow RN, Nester EW, Wood WA, Krieg NR, Phillips GB (eds). Manual of methods for general bacteriology. Washington, DC: American Society for Microbiology, pp 328–364
Heppel LA (1968) Preparation of cells ofEscherichia coli with altered permeability. Methods Enzymol 12B:841–846
Hisamatsu M, Yamada T, Higashiura T, Ikeda M (1987) The production of acidic, O-acylated cyclosophorans [cyclic(1–2)-beta-d-glucans] byAgrobacterium andRhizobium species. Carbohydr. Res. 163:115–122
Laimins LA, Rhoads DB, Epstein W (1981) Osmotic control ofkdp operon expression inEscherichia coli. Proc Natl Acad Sci USA 78:464–468
Lall SD, Eribo BE, Jay JM (1989) Comparison of four methods for extracting periplasmic proteins. J Microbiol Methods 9:195–199
Miller KJ, Kennedy EP, Reinhold VN (1986) Osmotic adaptation by gram-negative bacteria: possible role for periplasmic oligosaccharides. Science 231:48–51
Miller KJ, Reinhold VN, Weissborn AC, Kennedy EP (1987) Cyclic glucans produced byAgrobacterium tumefaciens are substituted withsn-1-phosphoglycerol residues. Biochim Biophys Acta 901:112–118
Miller KJ, Gore RS, Benesi AJ (1988) Phosphoglycerol substituents present on the cyclic beta-1,2-glucans ofRhizobium meliloti 1021 are derived from phosphatidylglycerol. J Bacteriol 170:4569–4575
Miller KJ, Gore RS, Johnson R, Benesi AJ, Reinhold VN (1990) Cell-associated oligosaccharides ofBradyrhizobium spp. J Bacteriol 172:136–142
Paoletti LC, Short KA, Blakemore N, Blakemore RP (1987) Freeze-thawing ofAquaspirillum magnetotacticum cells selectively releases periplasmic proteins. Appl Environ Microbiol 53:2590–2592
Puvanesarajah V, Schell FM, Stacey G, Douglas CJ, Nester EW (1985) Role for 2-linked-beta-d-glucans in the virulence ofAgrobacterium tumefaciens. J Bacteriol 164:102–106
Shapiro B, Stadtman ER (1970) Glutamine synthetase (Escherichia coli). Methods Enzymol 17A:910–922
Streeter JG (1989) Analysis of periplasmic enzymes in intact cultured bacteria and bacteroids ofBradyrhizobium japonicum andRhizobium leguminosarum biovarphaseoli. J. Gen. Microbiol. 135:3477–3484
Tully RE, Keister DL, Gross KC (1990) Fractionation of the beta-linked glucans ofBradyrhizobium japonicum and their response to osmotic potential. Appl Environ Microbiol 56:1518–1522
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miller, K.J., Gore, R.S. Cyclic beta-1,6-1,3 glucans ofBradyrhizobium: Functional analogs of the cyclic beta-1,2-glucans ofRhizobium?. Current Microbiology 24, 101–104 (1992). https://doi.org/10.1007/BF01570905
Issue Date:
DOI: https://doi.org/10.1007/BF01570905