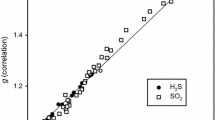
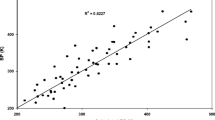
Abstract
We adapt existing models for estimating the second and third virial coefficients of small molecules to the halogenated methanes and ethanes. We compare the results with the abundant new, high-qualityPVT data resulting from the search for alternative refrigerants. The present model provides an accurate method for calculating densities, and therefore it should provide reliable thermodynamic properties and fugacity coefficients. We give equations and parameters useful for estimating the properties of pure refrigerants and their mixtures when noPVT data are available.
Similar content being viewed by others
Abbreviations
- B :
-
Second virial coeficient
- B 12 :
-
Mixture cross second virial
- B h :
-
Second virial of a hard sphere fluid
- ℬ i (T):
-
Temperature function, second virial, Eq. (7)
- C :
-
Third virial coefficient
- C 112,C 122 :
-
Mixture cross third virials
- C h :
-
Third virial of a hard sphere fluid
- N :
-
Avogadro's number
- P c :
-
Critical pressure
- P c12 :
-
Characteristic critical pressure of a binary mixture
- T c :
-
Critical temperature
- T c12 :
-
Characteristic critical temperature of a binary mixture
- T r :
-
Reduced temperature,T/T c
- α :
-
Parameter measuring polar contribution toB, Eq. (3)
- b :
-
Volume of a hard sphere molecule
- f (f) :
-
Polynomials determining temperature dependence of the nonpolar part ofB
- k 12 :
-
Binary interaction parameter for mixtures, Eq. (9a)
- χ c :
-
Critical volume
- α e :
-
Molecular polarizability
- μ :
-
Dipole moment
- μ R :
-
Reduced dipole moment, Eq. (4)
- μ R12 :
-
Mixture reduced dipole moment, second virial
- μ R112,μ R122 :
-
Mixture reduced dipole moment, third virial
- ω :
-
Pitzer acentric factor
- ω 12 :
-
Mixture acentric factor
- σ(r):
-
Intermolecular potential
- ϱ c :
-
Critical density (1/ηc)
References
S. C. Saxena and K. M. Joshi,Phys. Fluids 5:1217 (1962).
R. Schramm and C. H. Weber,J. Chem. Thermodyn. 23:281 (1991).
K. S. Pitzer and R. F. Curl,J. Am. Chem. Soc. 79:2369 (1957).
C. Tsonopoulos,AlChE. J. 20:263 (1974).
N. Van Nhu, G. A. Iglesias-Silva, and F. Kohler,Ber. Bunsenges. Phys. Chem. 93:526 (1989).
E. Bich, T. Lober, and I. Millat,Fluid Phase Equil. 75:149 (1992).
E. Bich, H. Hendle, T. Lober, and J. Millat,Fluid Phase Equil. 76:199 (1992).
C. Tsonopoulos,AlChE. J. 21:827 (1975).
J. P. O'Connell and J. M. Prausnitz,Ind. Eng. Chem. Process Des. Dev. 6:245 (1967).
J. H. Dymond and E. B. Smith,The Virial Coefficients of Pure Fluids and Mixtures (Clarendon Press, Oxford, 1980).
J. O. Hirschfelder, C. F. Curtiss, and R. B. Bird,Molecular Theory of Gases and Liquids (Wiley, New York, 1954), pp. 210–222.
W. H. Mears, R. F. Stahl, S. R. Orfeo, R. C. Shair, W. Thompson, and H. McCann,Ind. Eng. Chem. Ind. Ed. 47:1449 (1955).
R. Tillner-Roch and H. D. Baehr,J. Chem. Thermodyn. 24:413 (1992).
G. Morrison and M. O. McLinden,Int. J. Refrig. 16:129 (1993).
H. B. Lange and F. P. Stein,J. Chem. Eng. Data 15:56 (1970).
J. S. Rowlinson,J. Chem. Phys. 19:827 (1951).
P. L. Chueh and J. M. Prausnitz,AlChE. J. 13:896 (1967).
R. De Santis and B. Grande,AlChE. J. 25:931 (1979).
H. Orbey and J. H. Vera,AlChE. J. 29:107 (1983).
N. Van Nhu and F. Kohler,Ber. Bunsenges. Phys. Chem. 92:1129 (1988).
J. Gallagher, M. McLinden, and G. Morrison.NIST Thermodynamic Properties ol Refrigerants und Refrigerant Mixtures Database, Version 3.0, NIST Standard Reference Database 23 (National Institute of Standards and Technology, Gaithersburg, MD, 1992). (Copyright 1990, U.S. Department of Commerce)
L. A. Weber.Int. J. Thermophys. 10:617 (1989).
D. R. Deffibaugh, G. Morrison, and L. A. Weber. submitted for publication.
S. J. Boyes and L. A. Weber,Int. J. Thermophys. 15:443 (1994).
P. M. Sigmund, I. H. Silberberg, and J. J. McKetta,J. Chem. Eng. Data 17:168 (1972).
L. Haar and J. S. Gallagher,J. Phys. Chem. Ref. Data 7:635 (1978).
G. Natour, H. Schuhmacher, and B. Schramm,Fluid Phase Equil. 49:67 (1989).
G. Handel, R. Kleinrahm, and W. Wagner,J. ChEm. Thermodyn. 24:697 (1992).
C. M. Bignell and P. J. Dunlop.J. Chem. Eng. Data 38:139 (1993).
S. J. Boves and L. A. Weber, in press.
C. M. Bignell and P. J. Dunlop,J. Chem. Phys. 98:4889 (1993).
L. A. Weber,J. Chem. Eng. Data 35:237 (1990).
A. R. H. Goodwin and M. R. Moldover,J. Chem. Phys. 95:5236 (1991).
L. A. Weber,Proc. XVIII Int. Congr. Ref, Montreal (Int. Ins. Refr., Paris, 1991).
A. M. Demiriz, R. Kohlen, C. Koopmann, D. Moeller, P. Sauermann, G. A. Iglesias-Silva, and F. Kohler,Fluid Phase Equil. 85:313 (1993).
A. R. H. Goodwin and M. R. Moldover,J. Chem. Phys. 95:5230 (1991).
C. W. Meyer and G. Morrison,J. Phys. Chem. 95:3860 (1991).
A. R. H. Goodwin and G. Morrison,J. Phys. Chem. 96:5521 (1992).
L. A. Weber,Int. J. Thermophys. 13:1011 (1992).
H. B. Brugge, C.-A. Hwang, W. J. Rogers, J. C. Holste, K. R. Hall, W. Lemming, G. J. Esper, K. N. Marsh, and B. E. Gammon,Physica A 156:382 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weber, L.A. Estimating the virial coefficients of small polar molecules. Int J Thermophys 15, 461–482 (1994). https://doi.org/10.1007/BF01563708
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01563708