Abstract
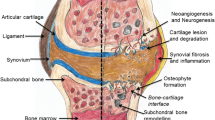
Approximately 44% of patients develop osteoarthritis (OA) following rupture of the anterior cruciate ligament (ACL) if the injury is left unrepaired. Restoring knee stability through reconstruction, while providing symptomatic relief, has not been shown to reduce the incidence of degenerative changes. In fact, recent studies have shown that 50%–60% of ACL-reconstructed patients go on to develop degenerative changes or frank osteo-arthritis. In light of these data, our group suggests that the cause of post-traumatic osteoarthritis is not biomechanical but biochemical. To test this hypothesis, we measured levels of nine cytokines which are important in modulating physiological and pathophysiological metabolism of cartilage in knee joint synovial fluid following ACL rupture. Our patient population contained both acute und chronic ACL ruptures. A total of 84 samples were collected and analyzed by enzyme-linked immunosorbent assay. On the basis of the data collected, we were able to identify subgroups of patients who, on the basis of their synovial fluid cytokine profile, may be at greater or leasser risk of developing post-traumatic OA. In general, patients displayed concentrations of interleukin-1α (IL-1α), basic fibroblastic growth factor (bFGF), transforming growth factor-β (TGF-β), granulocyte/macrophage-colony stimulating factor (GM-CSF), IL-6, and IL-8 that we interpreted as being consistent with an inflammatory reaction. Of great interest is the fact that the levels of these cytokines were very similar in patients 4 weeks after injury and in chronic patients, leading us to hypothesize that a chronic smoldering inflammatory reaction persists after resolution of the acute effusion. This chronic inflammatory reaction could be the cause of cartilage degeneration seen in patients after ACL reconstruction. Furthermore, this study identified two distinct subpopulations of patients with dramatically differing levels of IL-1β and TNF-α in their synovial fluids. IL-1β and TNF-α were either high acutely and decreased to moderate levels over time, or they were zero acutely and remained zero or low. This is likely to be a very important observation as these two cytokines are key components of the inflammatory response and are the main intra-articular cytokines that provoke cartilage loss. It is our hypothesis that the group with high levels of IL-1β and TNF-α contains the 50%–60% of the patient population which goes on to develop OA, despite knee stabilization with ACL reconstruction. Conversely, we suggest that the group whose synovial fluid contains no IL-1β or TNF-α is protected from developing OA post-traumatically. IL-1-receptor antagonist protein (IRAP), the cytokine that antagonizes the biological effects of IL-1, was present acutely in our patient population; however, it was undetectable in chronic ACL-deficient knees. These data suggest the loss with time of an important cartilage-protective cytokine. The lack of IRAP chronically may allow IL-1β to act in an unrestrained manner resulting in cartilage loss. In addition, IL-1β promotes the secretion of many other inflammatory cytokines which may result in the chronic smoldering inflammatory reaction referred to above. These data encourage further study of the evaluation of cytokine concentrations in the ACL-deficient joint as a means of obtaining both prognostic and mechanistic information of clinical value to the physician.
Similar content being viewed by others
References
Andrews HJ, Bunning RA, Dinarello CA, Russell RG (1989) Modulation of human chondrocyte metabolism by recombinant human interferon gamma: in vitro effects on basal and IL-1 stimulated proteinase production, cartilage degradation and DNA synthesis. Biochim Biophys Acta 102:128
Andrews HJ, Edwards TA, Cawston TE, Hazerman BL (1989) Transforming growth factor beta causes partial inhibition of interleukin-1 stimulated cartilage degradation in vitro. Biochem Biophys Res Commun 162:144
Arend WP, Smith MF, Janson RW, Joslin FG (1991) IL-1 receptor antagonist and IL-1β production in human monnocytes are regulated differently. J Immunol 147:1530–1536
Arner EC, Pratta MA (1989) Independent effects of interleukin-1 on proteoglycan breakdown, proteoglycan synthesis, and prostaglandin-E2 release from cartilage in organ culture. Arthritis Rheum 32:288–297
Bandara G, Lin CW, Georgescu HI, Evans CH (1992) Synovial activation of chondrocytes: evidence for complex cytokine interactions involving a possible novel factor. Biochim Biophys 1134:309–318
Bender S, Haubeck HD, Van de Leur E (1990) Interleukin-1β induces synthesis and secretion on interleukin-6 in human chondrocytes. FEBS Lett 263:321–324
Campbell IK, Piccoli DS, Roberts MJ (1990) Effects of tumor necrosis factor α and β on resorption of human articular cartilage and production of plasminogen activator by human articular chondrocytes. Arthritis Rheum 33:542–552
Carter DB, Beiberl MR, Dunn CJ (1990) Purification, cloning, expression and biological characterization of an interleukin-1 receptor antagonist protein. Nature 344:633–638
Chandrasekhar S, Harvey AK (1988) Transforming growth factor-β is a potent inhibitor of IL-1 induced protease activity and cartilage proteoglycan degradation. Biochem Biophys Res Commun 157:1352–1359
Dayer JM, Derochemonteix B, Burrus B (1986) Human recombinant interleukin-1 stimulates collagenase and prostaglandin E2 production by human synovial cells. J Clin Invest 77:645–648
Eisenberg SP, Evans RJ, Arend WP (1990) Primary structure and functional expression from complementary DNA of a human interleukin-1 receptor antagonist. Nature 343:341–346
Evans CH (1991) The role of proteinases in cartilage destruction. In: Parnham MJ, Bray MA, Van den Berg WB (eds) Drugs in inflammation. Birkhäuser, Basel, p 135
Ferretti A, Conteduca F, De Carli A, Fontana M, Mariani PP (1991) Osteoarthritis of the knee after ACL reconstruction. Int Orthop 15:367–371
Guerne PA, Zuraw BL, Vaughan JH, Carson DA, Lotz M (1989) Synovium as a source of interleukin-6 in vitro. J Clin Invest 83:585–592
Hawkins RJ, Misamore GW, Merritt TR (1986) Followup of the acute nonoperated isolated anterior cruciate ligament tear. Am J Sports Med 14:205–210
Hubbard JR, Steinberg JJ, Bednar MS, Sledge CB (1988) Effects of purified human interleukin-1 on cartilage degradation. J Orthop Res 6:180
Johnson RJ (1983) The anterior cruciate ligament problem. Clin Orthop 172:14–18
Kishimoto (1989) The biology of interleukin-6. Blood 74: 1–10
Larsen CG, Anderson AO, Appela E, Oppenheimer JJ, Matsushima K (1989) The neutrophil-activating protein (NAP-1) is also chemotactic for T-lymphocytes. Science 243:1464–1466
Le J, Vilcek J (1987) Biology of disease: tumor necrosis factor and interleukin-1: cytokines with multiple overlapping bioloical activities. Lab Invest 56:234–248
Le JM, Vilcek J (1989) Interleukin 6: a multifunctional cytokine regulating immune reactions and the acute phase protein response. Lab Invest 61:588–602
Locksley RM, Heinzel FP, Shephard HM (1987) Tumor necrosis factor-α and-β differ in their capacities to generate interleukin-1 release from human endothelial cells. J Immunol 139: 1891–1896
Lohmander LS, Dahlberg L, Ryd L, Heinegard D (1989) Increased levels of proteoglycan fragments in knee joint fluid after injury. Arthritis Rheum 32:1434–1442
Malyak M, Swaney RE, Arend WP (1993) Levels of synovial fluid interleukin-1 receptor antagonist in rheumatoid arthritis and other arthropathies: potential contribution from synovial fluid neutrophils. Arthritis Rheum 36:781–789
Martel-Pelletier J, Zafarullah M, Kodama S (1991) In vitro effects of IL-1 on the synthesis of metalloproteases, TIMP, plasminogen activators and inhibitors in human articular cartilage. J Rheumatol (Suppl) 27:80–84
Lefebure V, Peeters-Joris C, Vaes G (1990) Modulation of interleukin-1 and tumor necrosis factor-α of production of collagenase, tissue inhibitor of metalloproteinase and collagen types in differentiated and dedifferentiated articular chondrocytes. Biochim Biophys Acta 1052:366–378
Morales TI, Roberts AB (1988) Transforming growth factor-β regulates the metabolism of proteoglycans in bovine cartilage organ cultures. J Biol Chem 263:12828–12831
Morrissey PJ, Bressler L, Parks LS, Alpert A, Gillis S (1987) Granulocyte-macrophage colony stimulating factor augments the primary antibody response by enhancing the function of antigen-presenting cells. J Immunol 139:1113–1119
Murphy G, Hembry RM, Reynolds JJ (1986) Characterization of a specific antiserum to rabbit stromelysin and demonstration of the synthesis of collagenase and stromelysin by stimulated rabbit articular chondrocytes. Collagen Rel Res 6:351–364
Nietfeld JJ, wilbrink B, Helle M, Van Roy JL, Denooter W, Swaak AJ, Huber-Bruning O (1990) Interleukin-1 induced interleukin-6 is required for the inhibition of proteoglycan synthesis by interleukin-1 in human cartilage. Arthritis Rheum 33:1695
Noyes FR, Mooar PA, Matthews DS, Butler DL (1983) The symptomatic anterior cruciate-deficient knee. J Bone Joint Surg [Am] 65:154–162
Pratta MA, Di Meo TM; Ruhl DM (1989) Effects of interleukin-1 β and tumor necrosis factor-α on cartilage proteoglycan metabolism in vitro. Agents Actions 27:250–253
Pelletier JP, Roughley PJ, DiBattista JA, McCollum R, Pelletier JM (1991) Are cytokines involved in osteoarthritic pathophysiology? Arthritis Rheum 20:12–25
Pettipher ER, Higgs GA, Henderson B (1986) Interleukin-1 induces leukocyte infiltration and cartilage proteoglycan degradation in the synovial joint. Proc Natl Acad Sci USA 83:8749–8753
Rosenstreich DL, Tu JH, Kinkade PR (1988) A human urinederived interleukin-1 inhibitor. Homology with deoxyribonuclease I. J Exp Med 168:1767–1779
Saklatvala J (1986) Tumor necrosis factor-α stimulates resorption and inhibits synthesis of proteoglycan in cartilage. Nature 322:547–549
Roux-Lombard P, Punzi L, Hasler F, Bas S, Todesco S, Gallati H, Guerne P, Dayer J (1993) Soluble tumor necrosis factor receptors in human inflammatory synovial fluids. Arthritis Rheum 36:485–489
Seckinger P, Vey E, Turcatti G (1990) Tumor necrosis factor inhibitor: purification, NH2-terminal amino acid sequence and evidence for anti-inflammatory and immunomodulatory activities. Eur J Immunol 20:1167–1174
Seckinger P, Yaron I, Meyer FA, Yaron M, Dayer JM (1990) Modulation of the effects of interleukin-1 on glycosaminoglycan synthesis by the urine derived interleukin-1 inhibitor but not by interleukin-6. Arthritis Rheum 33:1807
Seckinger P, Williamson K, Balavoine JF (1987) A urine inhibitor of interleukin-1 activity affects both interleukin-1α and 1β but not tumor necrosis factor α. I Immunol 139:1541–1545
Smith RJ, Chin JE, Sam LM, Justen JM (1991) Biologic effects of interleukin-1 stimulated cartilage erosion and chondrocyte responsiveness. Arthritis Rheum 34:78–83
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cameron, M.L., Fu, F.H., Paessler, H.H. et al. Synovial fluid cytokine concentrations as possible prognostic indicators in the ACL-deficient knee. Knee Surg, Sports traumatol, Arthroscopy 2, 38–44 (1994). https://doi.org/10.1007/BF01552652
Issue Date:
DOI: https://doi.org/10.1007/BF01552652