Summary
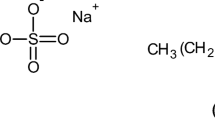
Measurements of the electrophoretic mobilities of the micelles of the anionic detergent C16H33(OCH2 · CH2)7OSO3Na have been made in aqueous sodium chloride solutions over the concentration range 0-0.01 M and used to calculate the magnitude of the electroviscous effect and the micellar charge in these solutions. The micellar hydration, as assessed from viscosity data, is shown to decrease as the salt concentration is increased and this is thought to be a consequence of the lower micellar charge in the more concentrated salt solutions which allows a greater contraction of the ethylene oxide chains resulting in a loss of water mechanically trapped by the micelles.
Similar content being viewed by others
References
Attwood, D., Kolloid-Z. u. Z. Polymere 232, 788 (1969).
Hoyer, H. W., K. J. Mysels and D. Stigter, J. Phys. Chem. 58, 385 (1954).
Booth, F., Proc. Roy. Soc. (London) A 203, 533 (1950).
Wiersema, P. H., A. L. Loeb and J. Th. G. Overbeek, J. Colloid Interfac. Sci. 22, 78 (1966).
Stigter, D. and K. J. Mysels, J. Phys. Chem. 59, 45 (1955).
Hunter, R. J., J. Phys. Chem. 66, 1367 (1962).
Stigter, D., Proc. 4th Inter. Congr. Surface-Active Substances, Brussels 2, 507 (1964).
Loeb, A. L., P. H. Wiersema and J. Th. G. Overbeek, The Electrical Double Layer Around a Spherical Particle (Cambridge, Mass. 1961).
Tokiwa, F., J. Phys. Chem. 72, 1214 (1968).
Tokiwa, F., J. Phys. Chem. 72, 4331 (1968).
Schott, H., J. Chem. Eng. Data 11, 417 (1966).
Tokiwa, F. and K. Ohki, J. Phys. Chem. 71, 1343 (1967).
Mukerjee, P., J. Colloid Sci. 19, 722 (1964).
Schick, M. J., J. Colloid Sci. 17, 801 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Attwood, D. The effect of electrolyte on the micellar charge and hydration of a polyoxyethylene sulphate type of detergent in aqueous solution. Kolloid-Z.u.Z.Polymere 235, 1193–1195 (1969). https://doi.org/10.1007/BF01542527
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01542527