Abstract
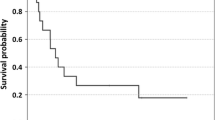
Chemotherapy and interleukin-2 (IL-2) and/or interferon α (IFNα) produce objective responses in a proportion of patients with advanced malignant melanoma. The duration of response to chemotherapy is usually less than 4 months, and immunotherapy has resulted in longlasting remissions in a small number of patients with metastatic melanoma. The current study was conducted to improve the antitumor efficacy and the interactions between recombinant (r) IL-2, rIFNα2a and chemotherapy. A total of 16 evaluable patients with metastatic malignant melanoma were entered into a phase-II study designed to assess the response rate and therapeutic efficacy of dacarbazine and carboplatin followed by rIL-2 and rIFNα2a. Patients received 750 mg/m2 dacarbazine with 400 mg/m2 carboplatin each by intravenous bolus on days 1 and 22. Recombinant IL-2 and IFNα2a were administered on an outpatient basis (home therapy) subcutaneously for 6 consecutive weeks: 4.8×106 IU/m2 rIL-2 daily, 5 days a week; 6.0×106 IU/m2 rIFNα2a thrice weekly. There were responses in 6 of the 16 enrolled patients with an overall response rate of 37.5% (95% confidence interval: 14%–61%). All responding patients had partial responses. The median survival time of the responding patients was significantly better than that of patients with progressive and stable disease (P=0.03). The median duration of response was 11 months (range 2–24 months). Responses in lung, liver, soft tissue and lymph-node sites were noted.
Similar content being viewed by others
References
Atzpodien J, Kirchner H (1990) Cancer, cytokines and cytotoxic cells: interleukin-2 in the immunotherapy of human neoplasms. Klin Wochenschr 68: 1
Atzpodien J, Krofer A, Evers P, Franks CR, Knuver-Hopf J, Fischer M, Mohr H, Dallmann I, Hadam M, Poliwoda H., Kirchner H (1990) Low-dose subcutaneous recombinant interleukin-2 in advanced human malignancy. A phase II outpatient study. Mol Biother 2: 18
Atzpodien J, Korfer A, Franks CR, Poliwoda H, Kirchner H (1990) Home therapy with recombinant interleukin-2 and interferon-α2b in advanced human malignancies. Lancet 335: 1509
Chang A, Hunt M, Parkinson DR, Hochster H, Smith TJ (1993) Phase II trial of carboplatin in patients with metastatic malignant melanoma. A report from the Eastern Cooperative Oncology Group. Am J Clin Oncol 16: 152
Coates A, Rallings M, Hersey P, Swanson C (1986) Phase II study of recombinant alpha-2 interferon in advanced malignant melanoma. J Interferon Res 6: 1
Creagan ET, Ahamann DL, Green SG, Long HG, Frytak S, O'Fullon JR, Itri LM (1984) Phase II study of low-dose recombinant leukocyte A interferon in disseminated malignant melanoma. J Clin Oncol 2: 1002
Eisenthal A, Skornick Y, Ron I, Zakuth V, Chaitchik S (1993) Phenotypic and functional profile of peripheral blood mononuclear cells isolated from melanoma patients undergoing combined immunotherapy and chemotherapy. Cancer Immunol Immunother 37: 367
Evans LM, Casper ES, Rosenbluth R (1987) Phase II trial of carboplatin in advanced malignant melanoma. Cancer Treat Rep 71:171
Falkson CI, Falkson G, Falkson HC (1991) Improved results with addition of interferon alfa-2b to dacarbazine in the treatment of patients with metastatic malignant melanoma. J Clin Oncol 9: 1403
Flaherty LE, Redman BG, Chabot GG, Martino S, Gualdoni SM, Heilbrun LK, Valdivieso M, Bradley EC (1990) A phase II study of dacarbazine in combination with outpatient interleukin-2 in metastatic malignant melanoma. Cancer 65: 2471
Flaherty LE, Robinson W, Redman BG, Gonzalez R, Martino S, Kraut M, Valdivieso M, Rudolph AR (1993) A phase II study of dacarbazine and cisplatin in combination with outpatient administered interleukin-2 in metastatif malignant melanoma. Cancer. 71: 3520
Fletcher J, Dana B, Jewell W, Townsend R, Fletcher W (1986) Evaluation of cisplatin and DTIC combination chemotherapy in disseminated melanoma: a SWOG phase II pilot study. Proc Am Soc Clin Oncol 5: 131
Goodnight JE, Moseley HS, Eilber FR, Sarna G, Morton DL (1979) Cis-dichlorodiammine-platinum (II) alone and combined with DTIC for treatment of disseminated malignant melanoma. Cancer Treat Rep 63: 2005
Gracomini P, Aguzzi A, Pestka S, Fisher PB, Ferrone S (1984) Modulation by recombinant DNA leukocyte (alpha) and fibroblast (beta) interferons by the expression and shedding of HLA- and tumor-associated antigens by human melanoma cells. J Immunol 133: 1649
Gresser I (1977) Commentary on varied biologic effects of interferon. Cell Immunol 34: 406
Hersey P, Hasic E, MacDonal M, Edwards A, Spurling A, Coates AS, Milton GW, McCarthy WH (1985) Effects of recombinant leukocyte interferon (r-IFN alpha A) on tumor growth and immune responses in patients with metastatic melanoma. Br J Cancer 51: 815
Hersey P, McLeod GRC, Thomson DB (1991) Treatment of advanced malignant melanoma with rIFNα in combination with DTIC. Br J Haematol 79: 60
Huberman M, Bering H, Fallon B, Tessitore J, Sonnenborn H, Paul S, Zeffren J, Levitt D, Groopman J (1991) A Phase I study of an outpatient regimen of recombinant human interleukin-2 and alfa-2a-interferon in patients with solid tumors. Cancer 68: 1708
Icli F, Karaguz H (1991) Treatment of metastatic malignant melanoma with 24 hours of continuous venous infusion of DTIC and cisplatin. J Surg Oncol 48: 199
Khavat D, Borel C, Antonie F, Benhamouda A, Soubrane CI, Auclerc G, Franks C, Weil M, Vuillemin E, Banzet P (1992) Highly active chemoimmunotherapy (cisplatin, IL-2, IFN-α) in the treatment of metastatic malignant melanoma. Proc ASCO 11: 332
Lee RE, Lotze MT, Skibber JM, Tucker E, Bonow RO, Ognibene FP, Carrasquillo JA, Shelhamer JH, Parrillo JE, Rosenberg SA (1989) Cardiorespiratory effects of immunotherapy with interleukin-2. J Clin Oncol 7: 7
Legha SS (1986) Interferons in the treatment of malignant melanoma, A review of recent trials. Cancer 57: 1675
Legha SS (1989) Current therapy for malignant melanoma. Semin Oncol 16: 34
McLeod GRC, Thomson DB, Hersey P (1987) rIFNα2a in advanced malignant melanoma, a phase I–II study in combination with DTIC. Int J Cancer 1 [Suppl]: 31
Oratz R, Speyer JL, Green M, Blum R, Wernz JC, Muggin FM (1987) Treatment of metastatic malignant melanoma with dacarbazine and cisplatin. Cancer Treat Rep 71: 877
Parkinson DR (1988) Interleukin-2 in cancer therapy. Semin Oncol 15: 10
Prichard KI, Quirt IC, Cowan DH, Osoba D, Kutas GJ (1980) DTIC therapy in metastatic malignant melanoma: a simplified dose schedule. Cancer Treat Rep 64: 1123
Ron IG, Inbar MJ, Gutman M, Merimsky O, Chaitchik S (1993) rIFNα2a in combination with dacarbazine in the treatment of metastatic malignant melanoma. Analysis of long-term responding patients. Cancer Immunol Immunother 37: 61
Rosenberg SA, Lotze MT, Muul LM, Chang AE, Avis FP, Leitman S, Linehan WM, Robertson CN, Lee RE, Rubin JT (1987) A progress report on the treatment of 157 patients with advanced cancer using lymphokine-activated killer-cells and interleukin-2 or high-dose interleukin-2 alone. N Engl J Med 316: 889
Rosenberg SA, Lotze MT, Yang JC, Aebersold PM, Lineban WM, Seipp CA, White DE (1989) Combination therapy with interleukin-2 and alpha-interferon for the treatment of patients with advanced cancer. J Clin Oncol 7: 1863
Rosenstein M, Ettinghausen SF, Rosenberg SA (1986) Extravasation of recombinant interleukin-2. J Immunol 137: 1735
Sertoli MR, Queirolo P, Bajetta E, Negretti E, Calabresi F, Bardugani L, Gianotti B, Cascinelli N (1992) DTIC with or without rIFNα2a at different dosages in the treatment of stage IV melanoma patients. Proc ASCO 11: 345
West WH, Taker KW, Yannelli JR, Marshall GD, Orr DW, Thurman GB, Oldham RK (1987) Constant infusion recombinant interleukin-2 in adoptive immunotherapy of advanced cancer. N Engl J Med 316: 898
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ron, H.G., Mordish, Y., Eisenthal, A. et al. A phase II study of combined administration of dacarbazine and carboplatin with home therapy of recombinant interleukin-2 and interferon-α2a in patients with advanced malignant melanoma. Cancer Immunol Immunother 38, 379–384 (1994). https://doi.org/10.1007/BF01517207
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01517207