Abstract
The composition and stability of complex ions of tri- and tetravalent plutonium in oxalate solutions were investigated polarographically.
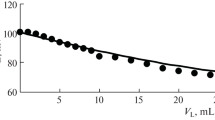
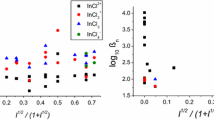
The complexes Pu(C2O4)4 −4 (preponderant amount) and Pu (C2O4)4 −5 were formed in solutions of potassium oxalate with pH 3.5–6. Under these conditions, Pu+4 gave a well expressed reverse reaction wave, suitable for the quantitative polarographic determination of plutonium. The oxidation-reduction potential of this reaction in 1 M potassium oxalate was equal to 0.205 v (relative to a saturated calomel electrode at a temperature of 25 ° C). Two Pu+4 complexes were simultaneously present in solutions at pH 6–8.
From data on the solubility of Pu(C2O4)3, we determined the instability constants of the complex oxalate ions Pu(C2O4)3 −3 and Pu(C2O4)4 −5, and from the polarographic data those forPu(C2O4)4 −4 ions:
Similar content being viewed by others
Literature cited
G. Seaborg and J. Katz, The Actinides (Foreign Lit. Press, 1955).
W. H. Reas, The Transuranium Elements (McGraw-Hill, 1949), 4, 14B, I, 423.
Beilsteins, Handbuch det Organischen Chemie 2, 477 (1942).
I. M. Kolthoff and D. D. Lingane, Polarography, (Foreign Lit, Press, 1945).
J. Heyrovsky and D. Ilkovic, Coll. Czech. Chem. Commun. 7, 198 (1935).
M. Stackelberg and H. Freyhold, Z. Elektrochem. 46, 120 (1940).
J. J. Lingane, Chem. Rev. 29, 1 (1941).
K. A. Kraus, The Transuranium Elements (McGraw-Hill, 1949) 4, 14B, 1, 241.
R. Brdicka, Coll. Czech. Chem. Commun. 2, 489, 545 (1930); 3, 396 (1931).
Rights and permissions
About this article
Cite this article
Fomin, V.V., Vorob'ev, S.P. & Andreeva, M.A. Polarographic investigation of complex plutonium oxalates. The Soviet Journal of Atomic Energy 4, 63–70 (1958). https://doi.org/10.1007/BF01514561
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01514561