Abstract
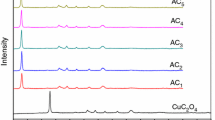
Decomposition of the oxalates of tri-, tetra- and hexavalent plutonium was studied in air and in a vacuum at room temperature and −80 both under illumination and in darkness. It was found that the decomposition is caused by alpha radiation from the plutonium, but in the oxalates of tetra- and hexavalent plutonium the carbon monoxide which is formed acts as a reducing agent which transforms the tetravalent plutonium to the trivalent form and the hexavalent to die tetravalent. The oxalates are then transformed into carbonates and, apparently, also partially into oxides or an oxypxalate-catbonate mixture.
Similar content being viewed by others
Literature cited
M. O. Kotshun and N. E. Gelman, New Methods of Elementary Microanalysis, State Tech. Press, 1949.
Bertold Stech, Z. Naturforsch. 2, 175 (1952).
S. L. Penfield, Am. J. Sci. (3) 48, 30 (1894).
A. G. Berg, Factory Lab. 10, 1171 (1948).
Rights and permissions
About this article
Cite this article
Fomin, V.V., Kartushova, R.E. & Rudenko, T.I. Decomposition of plutonium oxalates by intrinsic alpha radiation. The Soviet Journal of Atomic Energy 1, 409–413 (1956). https://doi.org/10.1007/BF01514509
Issue Date:
DOI: https://doi.org/10.1007/BF01514509