Abstract
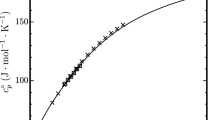
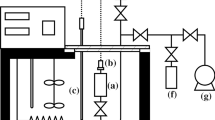
A fundamental equation ofstale for HFC-152a ( 1,1-dilluorocthane) is presented covering temperatures between the triple-point temperature ( 154.56 K) and 435 K for pressures up to 311 M Pa. The equation is based on reliable (p, g, T) data in the range mentioned above. These are generally represented within ±0.1 % of density. Furthermore. experimental values of the vapor pressure, the saturated liquid density, and some isobaric heat capacities in the liquid were included during the correlation process. The new equation of state is compared with experimental data and also with the equation of state developed by Tamatsu et al. Differences between the two equations of state generally result from using different experimental input data. It is shown that the new equation of state allows an accurate calculation of various thermodynamic properties for most technical applications.
Similar content being viewed by others

References
R. Tillner-Roth,Thermodynamie Properties of R 134a. R 153a and Their Mixtures—Meusurements and Fundamental Equations of State, Forsch.-Ber. DKV Nr. 41 (DKV, Stuttgart, 1993) (in German).
T. Hozumi, T. Koga, H. Sato, and K. Watanabe,Int. J. Thermophys. 14:739 (1993).
R. Tillner-Roth and H. D. Baehr.J. Chem. Thermodyn. 25:277 (1993).
R. Tillner-Roth and H. D. Baehr,J. Chem. Thermodyn. 24:413 (1992)
W. Blanke and R. Weiss,Fluid Phase Equil. 80:179 (1992).
M. Dressner and K. Bier,Thermal Mixing Effects in Binary Gaseous Mixtures of New Refrigerants, Fortschr.-Ber. VDI, Series, No. 332 (Düsseldorf, 1993) (in German).
E. G. Porichanskii, O. P. Ponomareva, and P. I. Svetlichnyi.Izv. Vys. Ucheb. Zaved. Energ. 3:122 (1982).
H. D. Baehr and R. Tillner-Roth,J. Chem. Thermodyn. 23:1063 (1991).
M. R. Moldover, J. P. M. Trusler, T. J. Edwards, J. B. Mehl, and R. S. Davis,J. Res. Natl. Bur. Stand. 93:85 (1988).
Y. Higashi, M. Ashizawa, Y. Kabata, T. Majima, M. Uematsu, and K. Watanabe,JSME Int. J. 30:1106 (1987).
W. Wagner,A Statistical Method JLr Establishing Thermodynamic Equations of State-Applied to the Vapour Pressure Curve of Pure Substances, Fortschr.-Ber. VDI. Series 3, No. 39 (Düsseldorf. 1974) (in German).
J. Ahrendts and H. D. Baehr,Int. J. Chem. Eng. 21:557 (1981).
T. Tamatsu, H. Sato, and K. Watanabe,Int. J. Refrig. 16:347 (1993).
T. Majima, M. Uematsu, and K. Watanabe,Proc. 8th Jap. Symp. Thermophys. Prop. 8:77 (1987).
T. Tarnatsu, T. Sato, H. Sato, and K. Watanabe,Int. J. Thermophys. 13:985 (1992).
Z. Y. Zhao, J. M. Yin, and L. C. Tan,Fluid Phase Equil. 80:191 (1993).
V. Z. Geiler, E. G. Porichanskii. P. I. Svetliclinyi, and Y. G. Elkin,Kholod. Tekh. 29:43 (1979).
A. Iso and M. Uematsu,Physica A 156:454 (1989).
S. Nakaeawa, T. Hori, H. Sato, and K. Watanabe,J. Chem. Eng. Data 38:70 (1993).
H. Kubota, H. Sugitani, Y. Tanaka, and T. Makita,Chemistry Express Kinki Chemical Society, Japan, 1987). Vol. 2, p. 397.
M. Takahashi. C. Yokoyama, and S. Takahashi.Proc. 7th Jap. Symp. Thermophys. Prop. (1986), pp. 179–182.
K. Bier, personal communication. University of Karlsruhe (1990).
V. Z. Geller, L. G. Porichanskii, P. I. Svetliclinyi, and Y. G. Elkin,Kholod. Tekh. 2:42 (1980).
H. Sato, M. Uematsu, and K. Watanabe,Fluid Phase Equil. 36:167 (1987).
J. Wang, Z. G. Liu, L. C. Tan and J. M. Yin,Fluid Phase Equil. 80:203 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tillner-Roth, R. A fundamental equation of state for 1,1-difluoroethane (HFC-152a). Int J Thermophys 16, 91–100 (1995). https://doi.org/10.1007/BF01438960
Issue Date:
DOI: https://doi.org/10.1007/BF01438960