Abstract
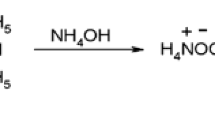
Reactions ofmyo-inositiol hexa-O-nitrate with ammonia and primary amines yield tetrahydroxy-1,4-benzoquinone derivatives,viz., its tetraammonium salt and its diimines, respectively. Reactions with secondary and tertiary amines give salts of rhodizonic acid, which are converted into salts of croconic acid under certain conditions. The reactions with secondary amines involve intermediate formation of radical species, which were dectected by ESR spectroscopy. A scheme for the chemical transformations ofmyo-inositol hexa-O-nitrate under the action of amines was proposed.
Similar content being viewed by others
References
Comprehensive Organic Chemistry, Eds. D. Barton and W. D. Ollis, Pergamon Press, Oxford 1979, 2.
K. Nakanishi,Infrared Absorption Spectroscopy, HoldenDay, Inc., San Francisco-Tokyo, 1962.
R. West,J. Am. Chem. Soc., 1960,82, 6204.
Z. Sharshenalieva, F. V. Pishugin, G. L. Rosenthal, and M. J. Strauss,Tetrahedron Lett., 1990,31, 1797.
R. West, inOxocarbon, Academic Press, New York, 1980, 3.
D. G. Cram,Fundamental Carbanion Chemistry, Academic Press, New York-London, 1965.
USSR Pat. 1747433,Byul. izobret., 1992, 26 (in Russian).
F. A. Hoglan and E. Barrow,J. Am. Chem. Soc., 1940,62, 2397.
A. J. Fatiadi, H. S. Isbell, and W. F. Sager,J. Res. Natl. Bur. Stand., 1963,A67, 153.
S. Ya. Pshezhetskii,EPR svobodnykh radikalov v radiatsionnoi khimii [ESR of Free Radicals in Radiation Chemistry], Khimiya, Moscow, 1972, 480 pp. (in Russian).
Yu. A. Zhdanov,Praktikum po khimii uglevodov [Laboratory Course on the Chemistry of Carbohydrates], Vysshaya shkola, Moscow, 1973, 84 pp, (in Russian).
A. Gordon and R. Ford,The Chemist's Companion, Wiley, New York, 1972.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 12, pp. 2962–2967, December, 1996.
Rights and permissions
About this article
Cite this article
Kuznetsov, Y.E., Sukhov, N.L., Pichugin, F.V. et al. Transformations ofmyo-inositol hexa-O-nitrate under the action of amines. Russ Chem Bull 45, 2816–2819 (1996). https://doi.org/10.1007/BF01430650
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01430650