Summary
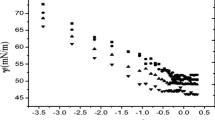
Surface and micellar properties of a homologous series of Octaethylene glycol-n-alkyl ethers (C n E8;n = 9 to 15) have been studied in aqueous solutions by the surface tension measurements. The effects of the alkyl chain length comprising even and carbon numbers have been examined in order to evaluate the surface free energy ΔG A-W and the standard free energy ΔG m for the micellization obtained from their surface tension data. The areas per molecule and the equilibrium surface tension values at the CMC decreased with an increasing carbon number and they showed zigzag curves by the difference in even and odd carbon numbers.
These findings may be attributed to the differences in the molecular orientation between the molecules with even carbon number and ones with odd carbon number on the air-water interface at CMC. ΔG A-W values decreased linearly with an increasing alkyl chain length but did not show a zigzag line by the differences in even and odd carbon numbers. This suggests that the molecular orientation is not influenced by the difference between their even and odd carbon numbers in the alkyl chain on the surface of the very diluted solution, and their molecules form some stable adsorbed films with an increase of the alkyl chain length. A division of ΔG A-W into the contribution made both by the hydrophilic group ΔG A-W (-W) and by the hydrophobic group ΔG A-W (-CH2-) was attempted as follows; ΔG A-W (-CH2-) = − 0.80 kcal/mol and ΔG a-W (-W) = + 0.15 kcal/mol.
The free energy changes ΔG m of micellization were discussed on the basis of the CMC data obtained from the surface tension measurements by treating the formation of micelles as analogous to phase separation, and the contribution from the each moieties in the molecule were calculated as follows; ΔG m (-CH2-) = − 0.68 kcal/mol and ΔG m (-W) = + 1.54 kcal/mol. The difference between ΔG A-W and ΔG m is discussed using their data.
Similar content being viewed by others
References
Corkill, J. M., T. F. Goodman, S. P. Harrold, Trans. Farady Soc.60, 202 (1964).
Carless, J. E., R. A. Challis, B. A. Mulley, J. Colloid Sci.19, 201 (1964).
Crook, E. H., D. B. Fordyce, G. F. Trebbi, J. Phys. Chem.67, 1987 (1963).
Schick, M. J., S. M. Atlas, F. R. Eirich, J. Phys. Chem.66, 1326 (1962).
Schick, M. J., E. A. Beyer, J. Amer. Oil Chem. Soc.40, 66 (1963).
Elworthy, P. H., A. T. Florence, Kolloid Z.195, 23 (1964).
Lange, H., J. Colloid Sci.20, 56 (1965).
Barry, B. W., D. I. D. El Eini, J. Colloid Interface Sci.54, 3 (1976).
Lange, H., Kolloid Z.201, 131 (1965).
Hsiao, L., H. N. Dunning, P. B. Lorenz, J. Phys. Chem.60, 657 (1956).
Becher, P., “Nonionic Surfactants”, P. 483. Academic Press, (New York, 1966).
Swarbrick, J., J. Pharm. Sci.58, 147 (1969).
Molyneux, P., C. T. Rhodes, J. Swarbrick, J. Trans. Farady Soc.61, 1043 (1965).
Traube, I., Liebigs Ann. Chem.265, 27 (1891).
Langmuir, I., J. Am. Chem. Soc.39, 1883 (1917).
Betts, J., B. Pethica, Proc. 2nd Intern. Congr. Surface Activity1, 152 (1957).
Weiner, N. D., G. Zografi, J. Pharm. Sci.54, 436 (1965).
Mcauliffe, C., Nature (London)200, 1092 (1963).
Wishnin, A., J. Phys. Chem.67, 2079 (1963).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ueno, M., Takasawa, Y., Miyashige, H. et al. Effects of alkyl chain length on surface and micellar properties of octaethyleneglycol-n alkyl ethers. Colloid & Polymer Sci 259, 761–766 (1981). https://doi.org/10.1007/BF01419322
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01419322