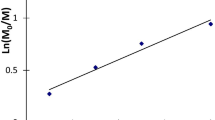
Abstract
Aqueous polymerization of acrylonitrile (M) initiated by the Ce(IV)-glucose (R) redox system has been studied under nitrogen in the temperature range of 30–40 °C. The rate of polymerization (Rp) is proportional to [M]2, [R] and inversely proportional to [Ce(IV)]. The rate of ceric ion disappearance is proportional to [R] and [Ce(IV)]. The end group in the polymer is characterised by IR spectra. A suitable kinetic scheme has been proposed and explained in the light of these experimental findings.
Similar content being viewed by others
References
Ananthanarayan, V. S., M. Santappa, J. Appl. Polym. Sci.9, 2437 (1965)
Saccubai, S., M. Santappa, J. Polym. Sci.A-l, 7, 643 (1969).
Devi, N. G., V. Mahadevan, Makromol. Chem.152, 177 (1972).
Nayak, P. L., R. K. Samal, M. C. Nayak, Eur. Polym. J.14, 287 (1978).
Singh, B. C., T. R. Mohanty, P. L. Nayak, J. Macromol. Sci. Chem.A-9, 1149 (1975).
Samal. R. K., B. C. Singh, T. R. Mohanty, P. L. Nayak, Makromol. Chem.176, 2987 (1975).
Wiberg, K. B. in: ‘Oxidation in Organic Chemistry’, Academic Press, Inc., New York (1965).
Rout, A., S. P. Rout, B. C. Singh, M. Santappa, Makromol. Chem.178, 639 (1977).
Rout, S. P., A. Rout, N. Mallick, B. C. Singh, M. Santappa, Makromol. Chem.178, 1971 (1977).
Bamford, C. H., A. D. Jenkins, Proc. Roy. Soc., LondonA 216, 515 (1953).
Mohanty, N., B. Pradhan, M. C. Mahanta, Eur. Polym. Journal15, 743 (1979).
Rout, A., S. P. Rout, N. Mallick, B. C. Singh, M. Santappa, J. Polym. Sci.16, 391 (1978).
Subramanian, S. V., M. Santappa, J. Poly. Sci. PartA-1, 6, 493 (1968).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Padhi, N.P., Singh, B.C. Vinyl polymerization of acrylonitrile by the Ce(IV)-glucose redox system. Colloid & Polymer Sci 261, 390–393 (1983). https://doi.org/10.1007/BF01418211
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01418211