Abstract
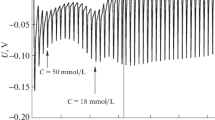
Phase transition temperatures from lyotropic liquid crystals to the isotropic micellar solution of the system cetyltrimethylammonium bromide-water (CTAB-H2O) were measured in the presence of 0.1-0.9 weight % of N-methyl-N,N-diphenylamine (MPA). They were determined by optical and viscometric methods and were found to increase as a function of MPA concentration in solutions containing 21 and 23 weight % of CTAB. This effect was reversed when MPA was in situ photochemically converted to N-methylcarbazole, allowing photochemically induced phase transitions.
Similar content being viewed by others
References
Wolff T, von Bünau G (1984) J Colloid Interface Sci 99:299
Wolff T, von Bünau G (1984) Ber Bunsenges Phys Chem 88:1098
Roessler N, Wolff T (1980) Photochem Photobiol 31:547
Wolff T, Suck TA, Emming C-S, von Bünau G (1987) Progr Colloid Polym Sci 73:18
Grellmann K-H, Kühnle W, Weller H, Wolff T (1981) J Am Chem Soc 103:6889
Götz KG (1961) Dissertation Universität Göttingen
Hertel G, Hoffmann H (1988) Progr Colloid Polym Sci 76:123
Sixl H (1984) Phys B1 40:35
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolff, T. Photochemically induced phase transitions in the system cetyltrimethyl-ammonium bromide-water containing N-methyl-N,N-diphenylamine. Colloid & Polymer Sci 267, 345–348 (1989). https://doi.org/10.1007/BF01413628
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01413628