Abstract
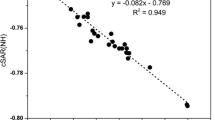
It is shown that the change of pKa of unsubstituted heterocyclic compounds on substitution is proportional to the difference of the residual π-electron charges at the position where the functional group is introduced in the unsubstituted heterobase and its conjugated acid.
Similar content being viewed by others
Literature cited
G. W. Wheland, “The quantum mechanics of unsaturated and aromatic molecules,” J. Chem. Phys.,2, No. 8, 474–481 (1934).
H. C. Longuette-Higgins, “Some studies in molecular orbital theory. 2. Ionization constants in heteroaromatic amines and related compounds,” ibid.,18, No. 3, 275–282 (1950).
S. F. Mason, “The tautomerism of N-heteroaromatic hydroxycompounds. 3. Ionization constants,” J. Chem. Soc., No. 2, 675–685 (1958).
R. Zahradnic and J. Koutecky, “Theoretical studies of physicochemical properties and reactivity of azines,” Adv. Heterocycl. Chem.,5, 69–118 (1965).
M. V. Bazilevskii, Molecular Orbital Methods and the Reactivity of Organic Molecules [in Russian], Khimiya, Moscow (1969).
V. F. Andriyanov, A. Ya. Kamenskii, S. S. Gitis, et al., “Infrared spectra and electronic effects. 4. The effect of substitution on the frequencies of the valence vibrations and the basicity of the amino groups in diphenyl systems with heterobridge,” Reakts. Sposobn. Org. Soedin.,12, No. 1, 91–101 (1975).
H. H. Jaffe, “Theoretical consideration concerning Hammett's equation. 4. Calculation of ρ-value,” J. Chem. Phys.,21, No. 3, 415–419 (1953).
R. Pones and V. Chralovsky, “Perturbation theory of substituent effect,” Coll. Czech. Chem. Commun.,11, No. 11, 3091–3099 (1974).
R. Pones, “Perturbation theory of substituent effect. Quantum chemical estimation of the Hammett ρ-constants,” ibid.,45, No. 6, 1646–1654 (1980).
Yu. B. Vysotskii, T. M. Prokop'eva, L. M. Litvinenko, et al., “Quantum chemical estimation of the transmitting ability of binuclear bridge systems,” Dokl. Akad. Nauk,234, No. 4, 854–857 (1977).
T. M. Prokop'eva, V. A. Dadali, Yu. B. Vysotskii, and L. M. Litvinenko, “Electronic structure and the transmitting ability in a series of bridge pyridine derivatives,” Zh. Org. Khim.,16, No. 6, 1207–1215 (1980).
V. A. Pal'ma (ed.), Tables of Rate and Equilibrium Constants of Heterolytic Organic Compounds [in Russian], Izd. Tart. Univ., Tart (1976).
A. Albert, “Ionization constants,” in: Physical Methods in Heterocyclic Chemistry, A. R. Katritzky (ed.), Vol. 1, Academic Press, New York (1963).
D. D. Perrin, Dissociation Constants of Organic Bases in Aqueous Solution, Butterworths, London (1965).
H. Sawada, M. Ichihara, Y. Yakawa, et al., “The substituent effect. 15. The-LSFE treatment of substituent effect on basicities of pyridine,” Bull. Chem. Soc. Jpn.,53, No. 7, 2055–2060 (1980).
M. Charton, “Substituent effects on pyridine nitrogen reactivity,” J. Org. Chem.,44, No. 13, 2097–2099 (1979).
G. Kortum, W. Vogel, and K. Andrussow, Dissoziations-Konstanten organischer SÄuren in Wasseriger Lösung, Butterworths, London (1961).
A. Albert, “Ionization constants,” in: Physical Methods in Heterocyclic Chemistry, Vol. 3, Academic Press, New York (1971), pp. 1–26.
H. H. Jaffe and G. O. Doak, “The basicities of substituted pyridines and their 1-oxides,” J. Am. Chem. Soc.,77, No. 20, 4441–4451 (1955).
A. K. Sheinkman, M. M. Mestechkin, A. P. Kucherenko, et al., “Reactions of cycloamine cations. 29. The effect of annelation on the reactivity of benzoquinolines in heteroannelation reactions,” Khim. Geterotsikl. Soedin., No. 8, 1096–1105 (1974).
Yu. B. Vysotskii and V. A. Sokolenko, “Electronic structure of excited states and spectroscopic properties of pyridine, quinoline, and their protonated forms,” Zh. Prikl. Spektrosk.,22, No. 4, 695–700 (1980).
Yu. B. Vysotskii, “The effect of chemical substitution on the ionization potential and electron affinity of systems with conjugated bonds,” Teor. Èsp. Khim.,17, No. 4, 469–478 (1981).
Author information
Authors and Affiliations
Additional information
Translated from Teoreticheskaya i éksperimental'naya Khimiya, Vol. 24, No. 1, pp. 13–20, January–February, 1988.
The author expresses his gratitude to M. M. Mestechkin and N. G. Korzhenevskaya for the discussion.
Rights and permissions
About this article
Cite this article
Vysotskii, Y.B. Quantum chemical study of the substituent effect on pKa of molecules with conjugated bonds using the PPP method. Theor Exp Chem 24, 12–18 (1988). https://doi.org/10.1007/BF01392184
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01392184