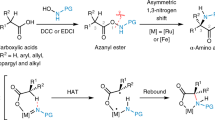
Summary
Various methodologies published in the literature dealing withα-amino carboxylic acid asymmetric synthesis are presented in a digest form. In each case, only some recent or most typical works are mentioned.
Similar content being viewed by others
References
Adger B, Berens U, Griffiths M, Kelly M, McCague R, Miller J, Palmer Ch, Roberts S, Selke R, Vitinius U, Ward G (1997) Chemoenzymatic synthesis of a novel ligand for rhodium-catalysed asymmetric hydrogenation. Chem Commun: 1713–1714
Aebi J, Seebach D (1985) Enantioselektiveα-Alkylierung von Asparagin- und Glutamins5ure über die Dilithium-Enolatecarboxylate von 2-[3-benzoyl-2-(tertbutyl)-1-methyl-5-oxoimidazolidin-4-yl]Essigsäure und 3-[3-benzoyl-2-(tert-butyl)-1methyl-5-oxoimidazolidin-4-yl]Propionsäure. Helv Chim Acta 68:1507–1518
Almena Perea J, Börner A, Knochel P (1998) A versatile modular approach to new chiral C2-symmetrical ferrocenyl ligands: highly Enantioselektive Rh-catalyzed hydrogenation ofα-acetamidoacrylic acid derivatives. Tetrahedron Lett 39: 8073–8076
Beckwith A, Chai C (1990) Diastereoselective radical addition to derivatives of dehydroalanine and of dehydrolactic acid. J Chem Soc Chem Commum: 1087–1088
Beylin V, Chen H, Dunbar J, Goel O, Harter W, Marlatt M, Topliss J (1993) Cyclic derivatives of 3,3-diphenylalanine (DIP) (II), novelα-amino acids for peptides of biological interest. Tetrahedron Lett 34: 953–956
Bravo P, Crucianelli M, Vergani B, Zanda M (1998) Sulfinimines of trifluoropyruvate: novel intermediates for chiral non racemicα-trifluoromethylα-amino acids. Tetrahedron Lett 39: 7771–7774
Burchat A, Chong J, Park S (1993) Observations on Sn-Li exchange inα-aminoorganostannanes and the configurational stability of non-stabilized aaminoorganolithiums. Tetrahedron Lett 34: 51–54
Calderari G, Seebach D (1985) Stereoselektive Alkylierung chiraler, nicht racemischer Enolate durch Nitroolefine. Herstellung enantiomerenreinerγ-Aminobuttersäure-und Bernsteinsaure-Derivate. Helv Chim Acta 68: 1592–1604
Calmes M, Daunis J, Mai N (1997a) Asymmetric synthesis ofα-amino acidsvia diastereoselective addition of (R)-pantolactone to their ketenes. Tetrahedron Asymm 8:1641–1648
Calmes M, Daunis J, Mai N (1997b) Tertiary amines: a decisive factor in the stereoselective addition of (R)-pantolactone to N-phthalyl valine ketene. Tetrahedron 53:13719–13726
Cardani S, Bernardi A, Colombo L, Gennari C, Scolastico C, Venturini I (1988) Asymmetric synthesis of functionalizedα-amino-β-hydroxy acids via chiral norephedrine-derived oxazolidines. Tetrahedron 44: 5563–5572
Cativiela C, Diaz-de-Villegas M, Galvez J (1992) Efficient asymmetric synthesis of amino acids through hydrogenation of the didehydroamino acid residue in cyclic imino-ester derivatives. Tetrahedron Asymm 3: 567–572
Chaari M, Jenhi A, Lavergne J-P, Viallefont Ph (1991) Synthese enantiosélective d'α-aryl amino acides: substitution nucléophile aromatique sur le fluorobenzène chrome tricarbonyle d'énolates chiraux. J Organomet Chem 401: C10–C13
Chai Ch, King A (1995) Methylene piperazine-2,5-diones as templates for the synthesis of amino acid derivatives. Tetrahedron Lett 36: 4295–4298
Chen H, Beylin V, Marlatt M, Leja B, Goel O (1992) Chiral synthesis of D- and L-3,3diphenylalanine (DIP), unusualα-amino acids for peptides of biological interest. Tetrahedron Lett 33: 3293–3296
Corey E, Xu F, Noe M (1997) A rational approach to catalytic enantioselective enolate alkylation using a structurallly rigidified and defined quaternary ammonium salt under phase transfer conditions. J Am Chem Soc 119: 12414–12415
Corey E, Noe M, Xu F (1998) Highly enantioselective synthesis of cyclic and functionalizedα-amino acids by means of chiral phase transfer catalyst. Tetrahedron Lett 39: 5347–5350
Davies J, Barrett G, Biagini S, Elmore D, Littlechild J, North M, Schofield C, Thirkettle J, Walter M, Williams P (1997) Amino acids and proteins, vol 28. A specialist periodical report. The Royal Society of Chemistry, Cambridge
Davis F, Fanelli D (1998) Sulfinimine-mediated asymmetric synthesis of (R)-(4-methoxy3,5-dihydroxyphenyl)glycine: the central aminoacid of vancomycin and related agents. J Org Chem 63: 1981–1985
Davis F, Reddy R, Portonovo P (1994a) Asymmetric Strecker synthesis using enantiopure sulfinimines: a convenient synthesis ofα-amino acids. Tetrahedron Lett 35: 9351–9354
Davis F, Zhou P, Reddy R (1994b) Asymmetric synthesis and reactions of cis-N(p-toluenesulfinyl)aziridine-2-carboxylic acids. J Org Chem 59: 3243–3245
Davis F, Portonovo P, Reddy R, Chin Y (1996a) Asymmetric Strecker synthesis using enantiopure sulfinimines and diethylaluminium cyanide: the alcohol effect. J Org Chem 61: 440–441
Davis F, Liu H, Reddy G (1996b) 2-MethylN-(p-toluenesulfinyl)aziridine-2-carboxylic acid: asymmetric synthesis ofα-methylphenylalanine andα-methyl-β-phenylserine. Tetrahedron Lett 37: 5473–5476
Davis F, Zhou P, Chen B-C (1998) Asymmetric synthesis of amino acids using sulfinimines (thiooxime S-oxides). Chem Soc Rev 27: 13–18
Decicco C, Grover P (1997) Asymmetric amino acid synthesis: Mitsunobu reaction on chiral cyanohydrins. Synlett: 529–530
Drury W, Ferraris D, Cox Ch, Young B, Lectka T (1998) A novel synthesis ofα-amino acid derivatives through catalytic, enantioselective ene reactions ofα-imino esters. J Am Chem Soc 120:11006–11007
Duhamel L, Plaquevent J-C (1978) Deracemization by enantioselective protonation. A new method for the enantiomeric enrichment ofα-amino acids. J Am Chem Soc 100: 7415–7416
Duhamel L, Plaquevent J-C (1980) Deracemization by enantioselective protonation IV: an improved method for the enantiomeric enrichment ofα-amino acids using metalation by means of chiral amides. Tetrahedron Lett 21: 2521–2524
Duhamel L, Duhamel P, Launay J-C, Plaquevent J-C (1984) Asymmetric protonations. Bull Soc Chim Fr 11: 421–430
Duhamel L, Fouquay S, Plaquevent J-C (1986) Ligand exchange in asymmetric reactions of lithium enolates: application to the deracemization ofα-amino acids. Tetrahedron 27:4975–4978
Duhamel L, Duhamel P, Fouquay S, Eddine J, Peschard O, Plaquevent J-C, Ravard A, Solliard R, Valnot J-Y, Vincens H (1988) Enantioselective electrophilic bond construction to theα-carbon ofα-amino acids. Tetrahedron 44: 5495–5506
Duthaler R (1994) Recent developments in the stereoselective synthesis ofα-amino acids. Tetrahedron 50: 1539–1650
Easton Ch (1997) Free-radical reactions in the synthesis ofα-amino acids and derivatives. Chem Rev 97: 53–82
Efferberger F, Burkard U, Wilfahrt J (1983) Trifluoromethanesulfonates ofα-hydroxy carboxylates — Educts for the racemization — Free synthesis of N-substitutedα-amino acid. Angew Chem Int Ed Engl 22: 65–66
El Achqar M, Boumzebra M, Roumestant M-L, Viallefont Ph (1988) 2-Hydroxy-3-pinanone as chiral auxiliary in the asymmetric synthesis ofα-amino acids. Tetrahedron 44: 5319–5332
Erdik E, Ay M (1989) Electropilic amination of carbanions. Chem Rev 89: 1947–1980
Evans D, Ellman J (1989) The total syntheses of the isodityrosine-derived cyclic tripeptides 0F4949-111 and K-13. Determination of the absolute configuration of K-13. J Am Chem Soc 111: 1063–1072
Evans D, Britton T, Dorow R, Dellaria J (1986) Stereoselective amination of chiral enolates. A new approach to the asymmetric synthesis ofα-hydrazino- andα-aminoacid derivatives. J Am Chem Soc 108: 6395–6397
Evans D, Britton T, Ellman J, Dorow R (1990a) The asymmetric synthesis ofα-amino acids. Electrophilic azidation of chiral imide enolates, a pratical approach to the synthesis of (R)- and (S)-α-azido carboxylic acids. J Am Chem Soc 112: 4011–4030
Evans D, Urpi F, Somers T, Clark J, Bilodeau M (1990b) New procedure for the direct generation of titanium enolates. Diastereoselective bond constructions with representative electrophiles. J Am Chem Soc 112: 8215–8216
Farr R (1998) Synthesis of chiral 3-substituted isoxazolines. Tetrahedron Lett 39: 195–196
Fehr Ch (1996) Enantioselective protonation of enolates and enols. Angew Chem Int Ed Engl 35: 2566–2587
Fiaud J-C, Kagan H (1970) Une nouvelle synthèse d'α-amino acides. Synthèse asymétrique de l'alanine. Tetrahedron Lett 21: 1813–1816
Fiaud J-C, Kagan H (1971) Synthèse asymétrique d'acides aminés. Etude de l'addition d'organometalliques sur les imines glyoxyliques. Tetrahedron Lett 22: 1019–1022
Fioravanti S, Loreto A, Pellacani L, Sabbatini F, Tardella P (1994) Asymmetric synthesis of N-(ethoxycarbon yl)-β-methylphenylat an in e esters. Tetrahedron Asymm 5: 473–478
Fryzuk M, Bosnich B (1977) Asymmetric synthesis. Production of optically active amino acids by catalytic hydrogenation. J Am Chem Soc 99: 6262–6267
Fuji K, Kawabata T (1998) Memory of chirality — A new principle in enolate chemistry. Chem Eur J 4: 373–376
Galvez N, Moreno-Manas M, Vallribera A, Molins E, Cabrero A (1996) Cobalt-mediated alkylation of (4R) and (4S)-3-acetoacetyl-4-benzyloxazolidin-2-ones. Preparation of enantiopure diphenylmethyl-, 9-fluorenyl- and (1-adanmantyl)glycines. Tetrahedron Lett 37: 6197–6200
Genêt J, Maillart S, Greek C, Pivereau E (1991) Electrophilic amination: first transfer of NHBoc with Li t-butyl-N-tosyloxycarbamate. Tetrahedron Lett 32: 2359–2362
Gennari C, Colombo L, Bertolini G (1986) Asymmetric Eecrophilic amination: synthesis ofα-amino acids andα-hydrazino acids with high optical purity. J Am Chem Soc 108: 6394–6395
Gittins C, North M (1997) Studies on the scope and applications of the catalysed asymmetric addition of organolithium reagents to imines. Tetrahedron Asymm 8: 3789–3799
Hamon D, Massy-Westropp R, Razzino P (1992) The asymmetric synthesis ofα-amino acids via the addition of Grignard reagents to imine derivatives. Tetrahedron 48: 5163–5178
Harada K (1963) Asymmetric synthesis ofα-amino acids by the Strecker synthesis. Nature 200: 1201
Harwood L, Tyler S, Anslow A, MacGilp I, Drew M (1997) Synthesis of enantiomerically pureα-amino acids via chemo- and diastereoselective alkylation of (5S)-5-phenyl-5,6-dihydro-2H-1,4-oxazin-2-one. Tetrahedron Asymm 8: 4007–4010
Hegedus L (1995) Synthesis of amino acids and peptides using chromium carbene complex photochemistry. Acc Chem Res 28: 299–305
Hegedus L, Lasrea E, Narukawa Y, Snustad D (1992) Asymmetric synthesis of (S)- and (R)-[2-2H1]glycine via photolysis of optically active chromium complexes: a comparison of stereoselectivity between chromium ketene complexes, ketenes, and ester enolates, J Am Chem Soc 114: 2991–2994
Hua D, Lagneau N, Wang H, Chen J (1995) Asymmetric synthesis ofα-amino acids via chiral N-alkylidenesulfinamides. Tetrahedron Asymm 6: 349–352
Iyer M, Gigstad K, Namdev N, Lipton M (1996) Asymmetric catalysis of the Strecker amino acid synthesis by a cyclic dipeptide. Amino Acids 11: 259–268
Jeaqjean F, Pérol N, Gore J, Fournet G (1997) Asymmetric carboxylation in the synthesis of L-methionine: a new tool for14C chemistry. Tetrahedron Lett 38: 7547–7550
Josien H, Chassaing G (1992) Asymmetric synthesis of the diastereoisomers of L-1indanylglycine and L-1-benz[f]indanylglycine, x1,x2-constrained side-chain derivatives of L-phenylalanine and L-2-naphthylalanine. Tetrahedron Asymm 3: 1351–1354
Jumnah R, Williams A, Williams J (1995) Synthesis of enantiomerically enrichedα-amino acid derivatives via transition metal catalysed procedures. Synlett: 821–822
Kagan H, Dang T (1972) Asymmetric catalytic reduction with transition metal complexes. I. A catalytic system of rhodium(I) with (−)-2,3-O-isopropylidene-2,3-dihydroxy-1,4-bis(diphenylphosphino)butane, a new chiral diphosphine. J Am Chem Soc 94: 6429–6433
Kawabata T, Wirth T, Yahiro K, Suzuki H, Fuji K (1996) Memory of chirality: direct asymmetricα-alkylation of phenylalanine derivatives. ICR Annu Rep 3: 36–37
Kober R, Papadopoulos K, Miltz W, Enders D, Steglich W (1985) Synthesis of diastereoand enantiomerically pureα-amino-γ-oxo acid esters by reaction of acyliminoacetates with enamines derived from 6-membered ketones. Tetrahedron 41: 1693–1701
Kochetkov K, Belikov V (1987) Modern asymmetric synthesis ofα-aminoacids. Russian Chem Rev 56: 1045–1067
Krause N, Ebert S, Haubrich A (1997) Diastereoselective protonation of chiral enolates with chelating proton donors under reagent control: scope, mechanism and applications. Liebigs Ann/Recueil: 2409–2418
Kreuzfeld H, Döbler Ch, Schmidt U, Krause H (1996) Synthesis of non-proteinogenic (D- or (L)-amino acids by asymmetric hydrogenation. Amino Acids 11: 269–282
Kubo A, Kubota H, Takahashi M, Nunami K-I (1996) Dynamic kinetic resolution utilizing 2-oxoimidazolidine-4-carboxylate as a chiral auxiliary: stereoselective synthesis ofα-amino acids by Gabriel reaction. Tetrahedron Lett 37: 4957–4960
Legters J, Willems J, Thijs L, Zwanenburg B (1992) Synthesis of functionalized amino acids by ring-opening reactions of aliphatically substituted aziridine-2-carboxylic esters. Reel Trav Chim Pays-Bas 111: 59–68
Liao S, Hruby V (1996) Asymmetric synthesis of optically pureβ-isopropyl phenylalanine: a newβ-branched unusual amino acid. Tetrahedron Lett 37: 1563–1566
Lundquist J, Dix T (1998) Asymmetric synthesis of\(\varpi \)-bromo-2(S)-azido acids as precursors for the synthesis of novel amino acids. Tetrahedron Lett 39: 775–778
Lygo B, Wainwright Ph (1997) A new class of asymmetric phase-transfer catalysts derived from Cinchona alkaloids - Application in the enantioselective synthesis ofα-amino acids. Tetrahedron Lett 38: 8595–8598
McIntoch J, Mishra P (1986) Alkylation of camphor imines of glycinates. Diastereoselectivity as a function of electronic factors in the alkylating agent. Canad J Chem 64: 726–731
McIntoch J, Leavitt R, Mishra P, Cassidy K, Drake J, Chadha R (1988) Diastereoselective alkylation guided by electrophile-nucleophileπ-interactions. J Org Chem 53: 1947–1952
Miyabe H, Ushiro C, Naito T (1997) Highly diastereoselective radical addition to glyoxylic oxime ether: asymmetric synthesis ofα-amino acids. Chem Comm: 1789–1790
Miyashita A, Yasuda A, Takaya H, Totiumi K, Ito T, Souchi T, Noyori R (1980) Synthesis of 2,2′-bis(diphenylphosphino)-1,1′-binaphthyl (BINAP), an atropisomeric chiral bis(triaryl)phosphine, and its use in the rhodium(I)-catalyzed asymmetric hydrogenation ofα-(acylamino)acrylic acids. J Am Chem Soc 102: 7932–7934
Morgan P, Whiting A, McCague R (1996) An asymmetric synthesis ofα-amino acid derivatives from racemic ethyl N-phenylsulphonyl-α-bromoglycinate using homochiral aluminium complexes. Tetrahedron Lett 37: 4795–4796
Myers A, Gleason J, Yoon T, Kung D (1997) Highly practical methodology for the synthesis of D- and L-α-amino acids and N-methyl-α-amino acids. J Am Chem Soc 119:656–673
O'Donnell M, Bennett W, Wu S (1989) The stereoselective synthesis ofα-amino acids by phase-transfer catalysis. J Am Chem Soc 111: 2353–2355
O'Donnell M, Delgado F, Hostettler C, Schwesinger R (1998) An efficient homogeneous catalytic enantioselective synthesis ofα-amino acid derivatives. Tetrahedron Lett 39: 8775–8778
Oppolzer W (1990) Camphor as a natural source of chirality in asymmetric synthesis. Pure Appl Chem 62:1241–1250
Oppolzer W, Tamura O (1990) Asymmetric synthesis ofα-amino acids andα-N-hydroxyamino acids via electrophilic amination of born anesultam-derived enolates with 1-chloro-1-nitrosocyclohexane. Tetrahedron Lett 31: 991–994
Oppolzer W, Pedrosa R, Moretti R (1986) Asymmetric syntheses ofα-amino acids fromα-halogenated 10-sulfonamido-isobornyl esters. Tetrahedron Lett 27: 831–834
Oppolzer W, Moretti R, Thomi S (1989) Asymmetric alkylations of a sultam-derived glycinate equivalent: practical preparation of enantiomerically pureα-amino acids. Tetrahedron Lett 30: 6009–6010
Osborn H, Sweeney J (1997) The asymmetric synthesis of aziridines. Tetrahedron Asymm 8: 1693–1715
Park Y, Beak P (1997) Enantioselective synthesis ofα-,β- andγ-aryl amino acids and esters. J Org Chem 62: 1574–1575
Patel M, Worsley M (1969) Asymmetric synthesis of optically activeα-amino acids by hydrocyanic acid addition to the optically active Schiff bases. Can J Chem 48: 1881–1884
Pellon P, Le Goaster C, Toupet L (1996) Diastereoselective synthesis of diphosphines, effect of their configuration in asymmetric catalysis. Tetrahedron Lett 37: 4713–4716
Phadtare S, Kamat S, Pause G (1985) Asymmetric synthesis ofα-amino acids. Ind J Chem 2413: 811–814
Porzi G, Sandri S (1996) Enantioselective synthesis of (R)- and (S)-α-amino acids using (6S)- and (6R)-6-methyl-morpholine-2,5-dione derivatives. Tetrahedron Asymm 7: 189–196
Pracejus G (1959) Optische Aktivierung von N-phthalyl-α-Aminosäure-Derivaten durch tert.-Basen-Katalyse. Liebigs Ann Chem 622: 10–22
Pye P, Rossen K, Reamer R, Tsou N, Volante R, Reider P (1997) A new planar chiral bisphosphine ligand for asymmetric catalysis: highly enantioselective hydrogenations under mild conditions. J Am Chem Soc 119: 6207–6208
Qian X, Russel K, Boteju L, Htuby V (1995) Stereoselective total synthesis of topographically constrained designer amino acids: 2′,6′-dimethyl-β-methyltyrosines. Tetrahedron 51: 1033–1054
RajanBabu T, Ayers T, Casalnuovo A (1994) Electronic amplification of selectivity in Rh-catalyzed hydrogenations: D-glucose-derived ligands for the synthesis of D- or Lamino acids. J Am Chem Soc 116: 4101–4102
Sabelle S, Lucet D, Le Gall Th, Miokowski Ch (1998) Enantioselective synthesis ofα-amino acids from nitroalkenes. Tetrahedron Lett 39: 2111–2114
Schlosser M, Limat D (1995) Sparteine-mediatedα-lithiation of N-Boc-N-methyl benzyl amine: rapid racemization and subsequent deracemization. J Am Chem Soc 117: 12342–12343
Schöllkopf U, Hartwig W, Groth U (1979) Enantioselective synthesis ofα-methyl-α-aminocarboxylic acids by alkylation of the lactim ether of cyclo-(L-Ala-L-Ala). Angew Chem Int Ed Engl 18: 863–864
Schöllkopf U, Groth U, Deng C (1981) Enantioselective synthesis of (R)-amino acids using L-valine as chiral agent. Angew Chem Int Ed Engl 20: 798–799
Schöllkopf U, Hupfeld B, Kuper S, Egert E, Dyrbusch M (1988) Enantioselective synthesis of (R)-N-Boc-l-amino-2-arylcyclopropene-l-carboxylic acid methyl ester. Angew Chem Int Ed Engl 27:433–434
Seebach D, Naef R (1981) Enantioselective generation and diastereoselective reactions of chiral enolates derived fromα-heterosubstitued carboxylic acids. Helv Chim Acta 64:2704–2708
Seebach D, Boes M, Naef R, Schweizer B (1983) Alkylation of amino acids without loss of the optical activity: preparation ofα-substituted proline derivatives. A case of selfreproduction of chirality. J Am Chem Soc 105: 5390–5398
Seebach D, Aebi J, Naef R, Weber T (1985)α-Alkylation of amino acids without racemization. Preparation of either (S)- or (R)-α-methyldopa from (S)-alanine.. Helv Chim Acta 68: 144–154
Seebach D, Sting A, Hoffmann M (1996) Self-regeneration of stereocenters (SRS) — Applications, limitations, and abandonment of a synthetic principle. Angew Chem Int Ed Engl 35: 2708–2748
Södergren M, Alonso D, Bedekar A, Anderson P (1997) Preparation and evaluation of nitrene precursors (Phl - NSO2Ar) for the copper catalyzed aziridination of olefins. Tetrahedron Lett 38: 6897–6900
Solas D, Hale R, Patel D (1996) An efficient synthesis of N-α-Fmoc-4-(phosphonodifluoromethyl)-L-phenylalanine. J Org Chem 61: 1537–1539
Strecker A (1850) Über die künstlische Bildung der Milchsäure und einen neuen, den Glycocoll homologen Körper. Liebigs Ann Chem 75: 27–51
Tabcheh M, El Achqar A, Pappalardo L, Roumestant M-L, Viallefont Ph (1991) Alkylation and protonation of chiral Schiff bases: diastereoselectivity as a function of the nature of reactants. Tetrahedron 47: 4611–4618
Tabcheh M, Guibourdenche Ch, Pappalardo L, Roumestant M-L, Viallefont Ph (1998) Diastereoselective protonation of enolates of Schiff bases. Tetrahedron Asymm 9: 1493–1495
Tamura M, Harada K (1980) Asymmetric synthesis of aspartic acid by hydrogenation of optically active 1,4-oxazine derivative. Bull Chem Soc Jpn 53: 561–562
Tanaka K, Alm M, Watanabe Y, Fuji K (1996) Asymmetric synthesis of uncommon aamino acids by diastereoselective alkylations of a chiral glycine equivalent. Tetrahedron Asymm 7: 1771–1782
Tanner D (1994) Chiral aziridines — their synthesis and use in stereoselective transformations. Angew Chem Int Ed Engl 33: 599–619
Togni A, Breutel C, Schnyder A, Spindler F, Landert H, Tijani A (1994) A novel easily accessible ferrocenyldiphosphine for highly enantioselective hydrogenation, allylic alkylation, and hydroboration reactions. J Am Chem Soc 116: 4062–4066
Trimble L, Vederas J (1986) Amination of chiral enolates by dialkyl azodiformates. Synthesis ofα-hydrazino acids andα-amino acids. J Am Chem Soc 108: 6397–6399
Vidal J, Guy L, Stérin S, Collet A (1993) Electrophilic amination: preparation and use of N-Boc-3-(4-cyanophenyl)oxaziridine, a new reagent that transfers a N-Boc group to N- and C-nucleophiles. J Org Chem 58: 4791–4793
Vigneron J-P, Kagan H, Horeau A (1968) Synthèse asymétrique de l'acide aspartique optiquement pur. Tetrahedron Lett 19: 5681–5683
Voyer N, Roby J (1995) An expedient enantioselective synthesis of N-Boc-protected phenylsarcosine. Tetrahedron Lett 36: 6627–6630
Voyer N, Roby J, Chénard S, Barberis C (1997) Enantioselective synthesis of phenylglycines using (−) sparteine.s-BuLi complex. Tetrahedron Lett 38: 6505–6508
Walkup R, Cole D, Whittlesey B (1995) Silicon-containing amino acids and peptides. Asymmetric synthesis of (trialkylsilyl)alanines. J Org Chem 60: 2630–2634
Ward R, Pelter A, Goubet D, Pritchard M (1995) Diastereoselective synthesis of abromoamides leading to diastereomerically enrichedα-amino-,α-hydroxy- and athiocarboxylic acid derivatives. Tetrahedron Asymm 6: 93–96
Williams R (1989) Synthesis of optically activeα-amino acids. In: Balwin J, Magnus P (eds) Organic chemistry series, vol 7. Pergamon Press, Oxford
Williams R, Im M-N (1988) Asymmetric synthesis ofα-amino acids: comparaison of enolate vs. cation functionalization of N-Boc-5,6-diphenyl-2,3,5,6-tetrahydro-4H-1,4oxazin-2-ones. Tetrahedron Lett 29: 6075–6078
Williams R, Sinclair P, Zhai D, Chen D (1988) Practical asymmetric syntheses ofα-amino acids through carbon-carbon bond constructions on electrophilic glycine templates. J Am Chem Soc 110: 1547–1557
Winter S, Pracejus H (1966) Überα-phthalamido-Ketene. Chem Ber 99: 151–159
Xiang L, Wu H, Hruby V (1995) Stereoselective synthesis of all individual isomers of 3-methyl-2′,6′-dimethylphenylalanine. Tetrahedron Asymm 6: 83–86
Yamada S-I, Hashimoto S-I (1976) Asymmetric transamination from amino acid (III) asymmetric synthesis of phenylglycine by chimical transamination from optically active amino acids to benzaldehyde. Chemistry Lett: 921–926
Yamada S-I, Oguri T, Shioiri T (1976) Asymmetric synthesis ofα-amino acid derivatives by alkylation of a chiral Schiff base. J Chem Soc Chem Comm: 136–137
Yamamoto Y, Ito W, Maruyama K (1985) Enantio- and diastereo-selective synthesis of amino acids via the reaction of allylic boron compounds withα-imino-esters. J Chem Soc Chem Comm: 1131–1132
Yamamoto Y, Ito W, Maruyama K (1988) Studies on the reaction ofα-imino esters with organometallic compounds. Tetrahedron 44: 5415–5423
Yaozhong J, Changyou Z, Huri P (1989) Asymmetric synthesis VII: double stereodifferentiation in allylation of ketimines bearing two chiral auxiliaries. Application to enantioselective synthesis ofα-allylglycine. Synthetic Commun 19: 881–888
Yaozhong J, Guilan L, Changyou Z, Huri P, Lanjun W, Aiqiao M (1991) Asymmetric synthesis XII I: the stereocontrolled synthesis of (R)-α-amino acids via a double chiral induction. Synthetic Commun 21: 1087–1090
Yeh T-L, Liao C-C, Uang B-J (1997) Enantioselective synthesis ofα-amino acids from glycinet-butyl ester. Tetrahedron 53: 11141–11152
Zheng, Amstrong J, McWilliams Ch, Volante R (1997) Asymmetric synthesis ofα-amino acid derivatives via an electrophilic amination of chiral amide cuprates with Li tbutyl-N-tosyloxycarbamate. Tetrahedron Lett 38: 2817–2820
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Calmes, M., Daunis, J. How to build optically activeα-amino acids. Amino Acids 16, 215–250 (1999). https://doi.org/10.1007/BF01388170
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01388170