Summary
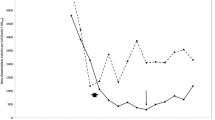
The production of penicillin amidase fromBacillus megaterium ATCC 14945 was examined in a sulfur-limited chemostat. Below 50% of the maximum growth rate, enzyme production was independent of dilution rate; above this point production decreased as growth rate increased. A simple and defined medium forBacillus licheniformis 749/C was developed, and extracellular penicillinase production was studied in a chemostat under sulfur- and nitrogenlimited conditions. While the yield of this enzyme differed little between these growth-limiting conditions, the specific activity of the penicillinase per unit weight of extracellular protein was increased 50% under nitrogenlimited conditions. Furthermore, the proportion of extracellular enzyme was greater in continuous, than in batch culture.
Similar content being viewed by others
References
Acevedo, F., Cooney, C.L. (1973). Biotechnol. Bioeng.15, 493–503
Ambler, R.P., Meadway, R.J. (1969). Nature22, 24–26
Borkowsky, J.D., Johnson, M.J. (1967). Biotechnol. Bioeng.9, 635–639
Chesbro, W.R., Lampen, J.O. (1968). J. Bacteriol.96, 428–437
Chiang, C., Bennett, R.E. (1967). J. Bacteriol.93, 302–308
Cooney, C.L. (1970). Double nutritional deficiencies in continuous microbial culture. Ph.D. thesis, Massachusetts Institute of Technology
Herbert, D., Elsworth, R., Telling, R.C. (1956). J. Gen. Microbiol.14, 601–622
Kirschenbaum, D.M. (1971). Anal. Biochem.44, 159–173
Lowry, O.H., Rosenbrough, N.J., Farr, A.L., Randall, R.J. (1951). J. Biol. Chem.193, 265–275
Murao,S., Sakaguchi,K., Kono,K. (1964). Process for preparing 6-APA byBacillus megaterium. U.S. Patent 3, 144, 395
Morihara, K. (1959). Bull. Agri. Chem. Soc. Japan23, 49–59
Niedermayer, A.O. (1964). Anal. Chem.36, 938–939
Oishi, M., Kitayama, S., Takahashi, H., Maruo, B. (1963). J. Gen. Appl. Microbiol.9, 337–341
Pollock, M.R., Richmond, M.H. (1962). Nature194, 446–449
Ryu, D.Y., Bruno, C.F., Lee, B.K. (1972). Microbial penicillin amidohydrolase and the performance of a continuous enzyme reactor system. In: Proceeding of the Fourth International Fermentation Symposium, pp. 307–314. Kyoto
Sargent, M.G. (1968). J. Bacteriol.95, 1493–1494
Sargent, M.G., Ghosh, B.K., Lampem, J.O. (1969). J. Bacteriol.97, 820–826
Slezak, J., Vojtisek, V., Sikyta, B. (1970). Penicillin acylase: regulatory mutants ofEscherichia colis. ATCC 9637. In: Genetics of industrial microorganisms, p. 78. Amsterdam/New York: Elsevier
Stickland, L.H. (1951). J. Gen. Microbiol.5, 698–703
Szentirmai, A. (1964). Appl. Microbiol.12, 185–187
Tomonaga, G. (1966). J. Gen. Appl. Microbiol.12, 267–276
Tomonaga, G., Ohama, H., Yamagita, T. (1964). J. Gen. Appl. Microbiol.10, 373–386
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Acevedo, F., Cooney, C.L. Penicillin amidase and penicillinase production in nitrogen- and sulfur-limited chemostats. European J. Appl Microbiol. 2, 9–17 (1975). https://doi.org/10.1007/BF01385441
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01385441