Abstract
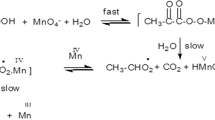
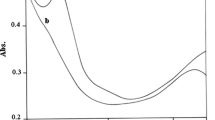
It was established that phosphoenolpyruvic acid, photoexcited by UV light at 20°C, loses its phosphoryl group under the action of acids and bases. Raising the temperature to 50°C induces dissociation of the photoexcited molecule into enolpyruvate and metaphosphate, which in turn phosphorylates HPO4 2− and H2P2O7 2− ions and AMP.
Similar content being viewed by others
References
K. Lohman and O. Meyerhoff, Biochem. Z.,273, No. 1, 560–572 (1934).
A. A. Yasnikov, N. P. Kanivets, A. L. Markovskii, and O. M. Lozinskaya, Zh. Obshch. Khim.,64, No. 2, 246–251 (1994).
D. Calvert and D. Piggs, Photochemistry [Russian translation], Mir, Moscow (1990).
F. H. Westheimer, Chem. Revs.,81, No. 4, 313–326 (1981).
D. Kuo, E. Connel, and I. Rose, J. Am. Chem. Soc.,101, No. 17, 5025–5030 (1979).
I. P. Beletskii and K. B. Yatsimirskii, Zh. Obshch. Khim.,56, No. 1, 35–40 (1986).
E. A. Shilov, Dokl. Akad. Nauk SSSR,18, No. 9, 643–648 (1938).
Additional information
Institute of Bioorganic Chemistry and Petrochemistry, National Academy of Sciences of Ukraine, 1 Murmanskaya St., 253094 Kiev-94, Ukraine. Translated from Teoreticheskaya i Éksperimental'naya Khimiya, Vol. 32, No. 5, pp. 287–290, September–October, 1996. Original article submitted December 1, 1995.
Rights and permissions
About this article
Cite this article
Yasnikov, A.A., Kanivets, N.P., Samus', N.V. et al. Kinetics and mechanism of the photochemical hydrolysis of phosphoenolpyruvic acid. Theor Exp Chem 32, 250–253 (1996). https://doi.org/10.1007/BF01384060
Issue Date:
DOI: https://doi.org/10.1007/BF01384060