Abstract
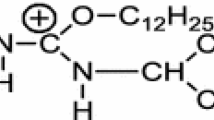
The absorption of cetyltrimethyl ammonium bromide onto silica was studied by FTIR as an example of the interaction of cationic surfactants with surfaces of medium polarity. It was found that the free silanol groups of the silica interacted strongly with the surfactant and that the methylene group adsorption bands varied with coverage in both frequency and width. These results could be interpreted in terms of chemisorption of a first layer of surfactant followed by physisorption of subsequent layers, or in terms of the inhomogeneity of the silica surface.
Similar content being viewed by others
References
J. M. Nicol,Ph. D. Thesis, University of Durham, 1985.
R. P. W. Scott, S. Traiman,J. Chromat. 1980,196, 193.
A. L. Dendramis, E. W. Schwinn, R. P. Sperline,Surface Science 1983,134, 675.
S. A. Savintseva, Z. A. Grankina, A. F. Koretskii,Kolloidnya Zhurnal 1976,38, 602.
R. K. Iler,Chemistry of Silica, Wiley-Interscience, New York, 1971.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sides, R., Yarwood, J. & Fox, K. An FTIR study of the adsorption of surfactants on silica. Mikrochim Acta 95, 93–96 (1988). https://doi.org/10.1007/BF01349728
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01349728