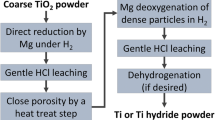
Abstract
Studies have been made on the kinetics of nickel oxide powder reduction by hydrogen in the presence of calcium oxide as a water vapor getter. The getter influences the reduction mechanism and the coalescence of the reduced metal as well as the growth of fresh surface. Data are presented on the fresh surface of the nickel powder formed in the presence of calcium oxide and in the absence of it as regards reduction kinetics.
Similar content being viewed by others
References
V. S. Zenkov, “Adsorption processes on getters for gaseous products from the reduction of metal oxides,” in: Current Advances in Physical Materials Science [in Russian], Inst. Probl. Materialovedeniya, NAN Ukrainy, Kiev (1995), pp. 42–55.
I. N. Frantsevich, V. S. Zenkov, V. N. Bulanov, et al., “The mechanism for the reaction of nickel oxide with hydrogen in the presence of catalytic amounts of Pd,” Zh. Fiz. Khim.,47, No. 9, 2206–2208 (1983).
N. A. Galaktionova, Hydrogen in Metals [in Russian], Metallurgizdat, Moscow (1967).
Additional information
Materials Science Institute, Ukrainian Academy of Sciences, Kiev. Translated from Poroshkovaya Metallurgiya, Nos. 5–6, pp. 79–83, May—June, 1996. Original article submitted June 29, 1994.
Rights and permissions
About this article
Cite this article
Zenkov, V.S., Uvarova, I.V. & Skorokhod, V.V. Effects of a getter on nio reduction kinetics and metallic nickel particle formation. Powder Metall Met Ceram 35, 286–289 (1996). https://doi.org/10.1007/BF01328835
Issue Date:
DOI: https://doi.org/10.1007/BF01328835