Summary
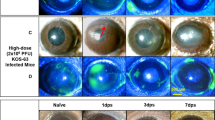
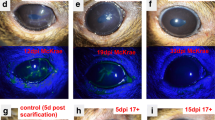
Herpes simplex virus can cause acute retinal necrosis, a blinding retinal disease in man. A unilateral intracameral inoculation of herpes simplex virus type 1 (HSV-1) in mice induces retinal necrosis primarily in the contralateral eye and provides an experimental model for the disease. Previous studies suggested that a major envelope glycoprotein of HSV-1, glycoprotein C (gC), is required for retinal necrosis. We studied HSV-1 strain TN-1, a gC-deficient clinical isolate from a lesion of herpetic keratitis, for its pathogenicity in mice with an intracameral inoculation of the virus and found that TN-1 could induce severe necrotizing retinitis in both inoculated and uninoculated eyes of BALB/c mice. Inoculation with a lower dose of TN-1 resulted in a unilateral necrotizing retinitis in the uninoculated eyes. Tissue virus titration of infected mice killed at various times after inoculation detected an infectious virus in various organs including the eyeballs, trigeminal ganglia, brain and adrenal glands. Anterior chamber-associated immune deviation (ACAID) was observed in TN-1-inoculated mice as well as in mice inoculated with gC-positive laboratory strain KOS 7 days postinoculation. Our findings suggested that gC of HSV-1 is not necessary for either the induction of retinal necrosis, neural spread of the virus, or ACAID.
Similar content being viewed by others
References
Atherton SS, Kanter MY, Streilein JW (1991) ACAID requires early replication of HSV in the injected eye. Curr Eye Res 10: 75–80
Atherton SS, Pesicka GA, Streilein JW (1987) Retinitis and deviant immune responses following intravitreal inoculation of HSV-1. Invest Ophthalmol Vis Sci 28: 859–866
Atherton SS, Streilein JW (1987) Two waves of virus following anterior chamber inoculation of HSV-1. Invest Ophthalmol Vis Sci 28: 571–579
Atherton SS, Streilein JW (1987) Virus-specific DTH prevents contralateral retinitis following intracameral inoculation of HSV-2. Curr Eye Res 6: 133–139
Culbertson WW, Blumenkranz MS, Pepose JS, Stewart JA, Curtin VT (1986) Varicella zoster virus is a cause of the acute retinal necrosis syndrome. Ophthalmology 93: 559–569
Glorioso J, Kees U, Kümel G, Kirchner H, Krammer PH (1985) Identification of herpes simplex virus type 1 (HSV-1) glycoprotein gC as the immunodominant antigen for HSV-1-specific memory cytotoxic T lymphocytes. J Immunol 135: 575–582
Hayashi Y, Nagata M, Yoshizumi H, Mori R, Navarro CV, Yang HD (1984) Difference in age distribution of antibody to herpes simplex virus in Fukuoka, Manila and Busan. Japan J Med Sci Biol 37: 35–39
Hemady R, Opremcak E, Zaltas M, Berger A, Foster C (1989) Herpes simplex virus type-1 strain influence on chorioretinal disease patterns following intracameral inoculation in Igh-1 disparate mice. Invest Ophthalmol Vis Sci 30: 1750–1757
Hendricks RL, Tao MSP, Glorioso JC (1989) Alterations in the antigenic structure of two major HSV-1 glycoproteins, gC and gB, influence immune regulation and susceptibility to murine herpes keratitis. J Immunol 142: 263–269
Hidaka Y, Sakai Y, Toh Y, Mori R (1991) Glycoprotein C of herpes simplex virus type 1 is essential for the virus to evade antibody-independent complement-mediated virus inactivation and lysis of virus-infected cells. J Gen Virol 72: 915–921
Hidaka Y, Sakuma S, Kumano Y, Minagawa H, Mori R (1990) Characterization of glycoprotein C-negative mutants of herpes simplex virus type 1 isolated from a patient with keratitis. Arch Virol 113: 195–207
Holland GN, Togni BI, Briones OC, Dawson CR (1987) A microscopic study of herpes simplex virus retinopathy in mice. Invest Ophthalmol Vis Sci 28: 1181–1190
Holland TC, Homa FL, Marlin SD, Levine M, Glorioso JC (1984) Herpes simplex virus type 1 glycoprotein C-negative mutants exhibit multiple phenotypes, including secretion of truncated glycoproteins. J Virol 52: 566–574
Igietseme JU, Streilein JW, Miranda F, Feinerman SJ, Atherton SS (1991) Mechanisms of protection against herpes simplex virus type 1-induced retinal necrosis by in vitro-activated T lymphocytes. J Virol 65: 763–768
Johnson RM, Lancki DW, Fitch FW, Spear PG (1990) Herpes simplex virus glycoprotein D is recognized as antigen by CD 4+ and CD 8+ T lymphocytes from infected mice. Characterization of T cell clones. J Immunol 145: 702–710
Kielty D, Cousins SW, Atherton SS (1987) HSV-1 retinitis and delayed hypersensitivity in DBA/2 and C 57BL/6 mice. Invest Ophthalmol Vis Sci 28: 1994–1999
Kumano Y, Yamamoto M, Inomata H, Sakuma S, Hidaka Y, Minagawa H, Mori R (1990) Recurrent herpetic keratitis: failure to detect herpes simplex virus infection using the Syva Microtrak HSV 1/HSV 2 direct specimen identification typing test. Ophthalmologica 201: 169–173
Langford MP, Colacino JM, Kaiwar R, Mahjoub SB, Ganley JP (1990) Cytopathogenicity, drug susceptibility, and thymidine kinase activity of a retinovirulent herpes simplex virus type 2. J Med Virol 31: 301–305
Lewis ML, Culbertson WW, Post MJD, Miller D, Kokame GT, Dix RD (1989) Herpes simplex virus type 1. A cause of the acute retinal necrosis syndrome. Ophthalmology 96: 875–878
Mahjoub SB, Ganley JP, Misra RP, Langford MP (1989) Isolation of a herpes simplex virus type 2 that is retinovirulent in mice. Curr Eye Res 8: 687–695
Martin S, Courtney RJ, Fowler G, Rouse BT (1988) Herpes simplex virus type 1-specific cytotoxic T lymphocytes recognize virus nonstructural proteins. J Virol 62: 2265–2273
Minagawa H, Sakuma S, Mohri S, Mori R, Watanabe T (1988) Herpes simplex virus type 1 infection in mice with severe combined immunodeficiency (SCID). Arch Virol 103: 73–82
Peiffer RL, Dekker CD, Siegel FL (1983) Ocular lesions in mice following intracerebral injection of herpes simplex virus type I. Invest Ophthalmol Vis Sci 24: 1070–1078
Rouse BT (1984) Cell-mediated immune mechanisms. In: Rouse BT, Lopez C (eds) Immunobiology of herpes simplex virus infection. CRC Press, Boca Raton, pp 107–120
Schreier RD, Pizer LI, Moorhead JW (1983) Type-specific delayed hypersensitivity and protective immunity induced by isolated herpes simplex virus glycoprotein. J Immunol 130: 1413–1418
Streilein JW, Atherton S, Vann V (1987) A critical role for ACAID in the distinctive pattern of retinitis that follows anterior chamber inoculation of HSV-1. Curr Eye Res 6: 127–131
Streilein JW, Niederkorn JY (1981) Induction of anterior chamber-associated immune deviation requires an intact, functional spleen. J Exp Med 153: 1058–1067
Szily A von (1924) Experimentelle endogene Infektionsübertragung von Bulbus zu Bulbus. Ein Beitrag zur Erforschung der sympathischen Ophthalmie und zur Pathogenese des hämatogenen Herpes corneae. Monatsbl Augenheilkd 72: 593–602
Toh Y, Tanaka S, Liu Y, Hidaka Y, Mori R (1993) Molecular characterization of naturally occurring glycoprotein C-negative herpes simplex virus type 1. Arch Virol 129: 119–130
Vann VR, Atherton SS (1991) Neural spread of herpes simplex virus after anterior chamber inoculation. Invest Ophthalmol Vis Sci 32: 2462–2472
Whitley RJ (1990) Herpes simplex viruses. In: Fields BN, Knipe DM (eds) Virology, vol 2, 2nd edn. Raven Press, New York, pp 1843–1887
Whittum JA, McCulley JP, Niederkorn JY, Streilein JW (1984) Ocular disease induced in mice by anterior chamber inoculation of herpes simplex virus. Invest Ophthalmol Vis Sci 25: 1065–1073
Whittum JA, Niederkorn JY, McCulley JP, Streilein JW (1982/1983) Intracameral inoculation of herpes simplex virus type I induces anterior chamber associated immune deviation. Curr Eye Res 2: 691–697
Witmer LA, Rosenthal KL, Graham FL, Friedman HM, Yee A, Johnson DC (1990) Cytotoxic T lymphocytes specific for herpes simplex virus (HSV) studied using adenovirus vectors expressing HSV glycoproteins. J Gen Virol 71: 387–396
Zaltas MM, Opremcak EM, Hemady R, Foster CS (1992) Immunohistopathologic findings in herpes simplex virus chorioretinitis in the von Szily model. Invest Ophthalmol Vis Sci 33: 68–77
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, Y., Sakai, Y., Minagawa, H. et al. Induction of bilateral retinal necrosis in mice by unilateral intracameral inoculation of a glycoprotein-C deficient clinical isolate of herpes simplex virus type 1. Archives of Virology 129, 105–118 (1993). https://doi.org/10.1007/BF01316888
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01316888