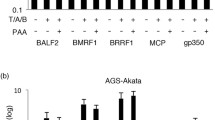
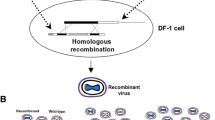
Summary
Lytic transition of Epstein-Barr virus (EBV) is initiated by distinct immediate early regulators of the viral cycle, in synchronization to temporary, permissive conditions during host cell differentiation. We developed eukaryotic vectors suitable to imitate the processes involved in lytic transition in cell culture systems. Two stable B cell lines were established: R59Z activator cells were used to induce lytic EBV expression in a constitutive manner by the production of the BZLF 1trans-activator (Zta). R7-57 reporter cells, on the other hand, signaled induced activity of the lytic origin of EBV replication (oriLyt). Different modes, like chemical induction, lytic superinfection with EBV and single genetrans-activation converted the recombinant oriLyt element in R7-57 reporter cells. BZLF 1, transiently expressed in R7-57 reporter cells, was the only EBVtrans-activator found, sufficient in inducing the viral lytic cycle. Basing on these experiments,trans-cellular activation of EBV was tested by cocultivation of BZLF 1-expressing R59Z activator cells with the R7-57 reporter line. No lytic effect on the reporter cells could be measured, neither by cocultivation of activator cells nor by coincubation of BZLF 1-containing cell lysates. Latency breaking activity, however, was transferred from activator to reporter cells when active, exogenous virus was added. The cell system described in these experiments provides a tool for the detection of EBV reactivation and demonstrates the potential of the lytic regulatory gene BZLF 1.
Similar content being viewed by others
References
Baer R, Bankier AT, Biggin MD, Deininger PL, Farell P, Gibson TJ, Hatfull G, Hudson GS, Satchwell SC, Seguin C, Tuffnell PS, Barrell BG (1984) DNA sequence and expression of the B 95-8 Epstein-Barr virus genome. Nature 310: 207–211
Becker J, Leser U, Marschall M, Langford A, Jilg W, Gelderblom H, Reichart P, Wolf H (1991) Expression of proteins encoded by Epstein-Barr viraltrans-activator genes depends on the differentiation of epithelial cells in oral hairy leukoplakia. Proc Natl Acad Sci USA 88: 8332–8336
Biggin M, Bodescot M, Pericaudet M, Farrell P (1987) Epstein-Bar virus gene expression in P3HR1-superinfected Raji-cells. J Virol 61: 3120–3132
Boshart M, Weber F, Jahn G, Dorsch-Häsler K, Fleckenstein B, Schaffner W (1985) A very strong enhancer is located upstream of an immediate early gene of human cytomegalovirus. Cell 41: 521–530
Countryman J, Jenson H, Seibl R, Wolf H, Miller G (1987) Polymorphic proteins encoded within BZLF 1 of defective and standard Epstein-Barr viruses disrupt latency. J Virol 12: 3672–3679
Countryman J, Miller G (1985) Activation of expression of latent Epstein-Barr herpesvirus after gene transfer with a small cloned subfragment of heterogeneous viral DNA. Proc Natl Acad Sci USA 82: 4085–4089
Cox MA, Leahy J, Hardwick MJ (1990) An enhancer within the divergent promoter of Epstein-Barr virus responds synergistically to the R and Z transactivators. J Virol 64: 313–321
Ensoli B, Barillari G, Salahuddin SZ, Gallo RC, Wong-Staal F (1990) Tat protein of HIV-1 stimulates growth of cells derived from Kaposi's sarcoma lesions of AIDS patients. Nature 345: 84–86
Flemmington E, Speck S (1990) Identification of phorbol ester response elements in the promoter of Epstein-Barr virus putative lytic switch gene BZLF 1. J Virol 64: 1217–1226
Hammerschmidt W, Sugden B (1988) Identification and characterization of oriLyt, a lytic origin of DNA replication of Epstein-Barr virus. Cell 55: 427–433
Hatfull G, Bankier AT, Barrel BG, Farrell PJ (1988) Sequence analysis of Raji Epstein-Barr virus DNA. Virology 164: 334–340
Garcia-Blanco MA, Cullen BR (1991) Molecular basis of latency in pathogenic human viruses. Science 254: 815–819
Gruffat H, Moremo N, Sergeant A (1990) The Epstein-Barr virus (EBV) ORIlyt enhancer is not B-cell specific and does not respond synergistically to the EBV transcription factors R and Z. J Virol 64: 2810–2818
Helland DE, Welles JL, Caputo A, Haseltine WA (1991) Transcellular transactivation by the human immunodeficiency virus type 1tat protein. J Virol 65: 4547–4569
Katz DA, Baumann RP, Sun R, Kolman JL, Taylor N, Miller G (1992) Viral proteins associated with the Epstein-Barr virus transactivator, ZEBRA. Proc Natl Acad Sci USA 89: 378–382
Laux G, Freese UK, Fischer R, Polack A, Kofler E, Bornkamm GW (1988) TPA-inducible Epstein-Barr virus genes in Raji cells and their regulation. Virology 162: 503–507
Liebermann PM, Berk AJ (1990) In vitro transcriptional activation, dimerization and DNA-binding specificity of the Epstein-Barr virus Zta protein. J Virol 64: 2560–2568
Lindholm PF, Reid RL, Brady J (1992) Extracellular Tax1 protein stimulates tumor necrosis factor-β and immunoglobulin kappa light chain expression in lymphoid cells. J Virol 66: 1294–1302
Loning T, Henke RP, Reichart R, Becker J (1987) In situ hybridisation to detect Epstein-Barr virus DNA in oral tissues of HIV-infected patients. Virchows Archiv 412: 127–133
Manet E, Gruffat H, Trescol-Biemont M, Moreno N, Chambard P, Giot J, Sergeant A (1989) Epstein-Barr virus bicistronic mRNAs generated by facultative splicing code for two transcriptional trans-activators. EMBO J 8: 1819–1826
Margolskee RF, Kavathas P, Berg P (1988) Epstein-Barr virus shuttle vector for stable episomal replication of cDNA expression libraries in human cells. Mol Cell Biol 8: 2837–2847
Marschall M, Motz M, Leser U, Schwarzmann F, Oker B, Wolf H (1989) Hepatitis B virus surface antigen as a reporter of promoter activity. Gene 81: 109–117
Marschall M, Schwarzmann F, Leser U, Oker B, Allinger P, Mairhofer H, Wolf H (1991) The BI'LF 4trans-activator of Epstein-Barr virus is modulated by type and differentiation of the host cell. Virology 181: 172–179
McCutchan JH, Pagano JS (1968) Enhancement of the infectivity of simian virus 40 deoxyribonucleic acid with diethylaminoethyl-dextran. J Natl Cancer Inst 41: 351–357
Miller G (1990) The switch between latency and replication of Epstein-Barr virus. J Infect Dis 161: 833–844
Montalvo EA, Shi Y, Shenk TE, Levine A (1991) Negative regulation of the BZLF 1 promoter of Epstein-Barr virus. J Virol 65: 3647–3655
Sambrook J, Fritsch E, Maniatis T (1989) Molecular cloning: a laboratory manual, second edition. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sato H, Takeshita H, Furukawa M, Seiki M (1992) Epstein-Barr virus BZLF 1 transactivator is a negative regulator of jun. J Virol 66: 4732–4736
Schuster C, Chasserot-Golat S, Beck G (1991) Activation of Epstein-Barr virus promoters by a growth-factor and a glucocorticoid. FEBS 284: 82–86
Skare J, Strominger JL (1980) Cloning and mapping of Bam HI endonuclease fragments of DNA from the transforming B 95-8 strain of Epstein-Barr virus. Proc Natl Acad Sci USA 77: 3860–3864
Takada K, Ono Y (1989) Synchronous and sequential activation of latently infected Epstein-Barr virus genomes. J Virol 63: 445–449
Yates JL, Warren N, Sugden B (1985) Stable replication of plasmids derived from Epstein-Barr virus in various mammalian cells. Nature 313: 812–815
Young LS, Lau R, Rowe M, Niedobotek G, Packham G, Shanahan F, Rowe DT, Greenspan D, Greenspan JS, Rickinson AB, Farrell PJ (1991) Differentiation-associated expression of the Epstein-Barr virus BZLF 1 transactivator protein in oral hairy leukoplakia. J Virol 65: 2868–2874
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marschall, M., Alliger, P., Schwarzmann, F. et al. The lytic transition of Epstein-Barr virus is imitated by recombinant B-cells. Archives of Virology 129, 23–33 (1993). https://doi.org/10.1007/BF01316882
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01316882