Abstract
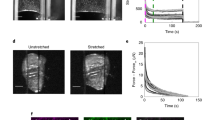
We have developed a model system to quantify the tractional forces generated by intestinal epithelial cells during organization into a confluent epithelial cell sheet. In this model system, IEC-6 cells, a rat intestinal crypt cell line, rapidly contracted collagen gels reducing the gel surface area by 97% at 24 hr. The tractional forces measured by gel contraction were directly related to the number of cells added and were inversely related to the collagen concentration of the gel. Actin microfilament function was required for gel contraction, but microtubular function was not. Fetal bovine serum and protein synthesis were required for maximal gel contraction. IEC-6 (5×105) cells per gel and fibroblasts (5 ×104) cells added to collagen gels resulted in contraction of the gels by 50% at 24 hr. Therefore, intestinal epithelial cells and fibroblasts generate tractional forces of similar strength capable of organizing the surrounding extracellular matrix, which should be considered in models of intestinal morphogenesis and repair.
Similar content being viewed by others
References
Kleinman HK, McGarvey ML, Hassell JR, Star VL, Cannon FB, Laurie GW, Martin GR: Basement membrane complexes with biological activity. Biochemistry 25:312–318, 1986
Klienman HK, McGarvey ML, Hassell JR, Martin GR, Baron von Evercooren A, Dubois-Dalccq M: The role of laminin in basement membranes and in the growth, adhesion, and differentiation of cells. The role of extracellular matrix in development. 123–143, 1984
Quaroni A: Development of fetal rat intestine in organ and monolayer culture. J Cell Biol 100:1611–1622, 1985
Olson AD, Pysher T, Bienkowski RS: Organization of intestinal epithelial cells into multicellular structures requires laminn and functional actin microfilaments. Exp Cell Res 192:in press, 1991
Dunn G, Heath J: A new hypothesis of contact guidance in tissue cells. Exp Cell Res 101:1–14, 1976
Dunn G, Ebendal T: Contact guidance on oriented collagen gels. Exp Cell Res 111:475–479, 1978
Elsdale T, Bard J: Collagen substrata for studies on cell behavior. J Cell Biol 54:626–637, 1972
Farsi JMA, Aubin JE: Microfilament rearrangements during fibroblast-induced contraction of three-dimensional hydrated collagen gels. Cell Motil 4:29–40, 1984
Guidry C, Grinnell F: Contraction of hydrated collagen gels by fibroblasts: Evidence for two mechanisms by which collagen fibrils are stabilized. Collagen Relat Res 6:515–529, 1987
Fukamizu H, Grinnell F: Spatial organization of extracellular matrix and fibroblast activity: Effects of serum, transforming growth factor beta, and fibronectin. Exp Cell Res 190:276–282, 1990
Grinnell F, Fukamizu H, Pawelek P, Nakagawa S: Collagen processing, crosslinking, and fibril bundle assembly in matrix produced by fibroblasts in long-term cultures supplemented with ascorbic acid. Exp Cell Res 181:483–491, 1989
Schor S, Schor A: Foetal-to-adult transitions in fibroblast phenotype: Their possible relevance to the pathogenesis of cancer. J Cell Sci Suppl 8:165–180, 1987
Allan T, Schor S: The contraction of collagen matrices by dermal fibroblasts. J Ultrastruct Res 83:205–219, 1983
Bell E, Ivarsson B, Merrill C: Production of a tissue-like structure by contraction of collagen lattices by human fibroblasts of different proliferative potentialin vitro. Proc Natl Acad Sci USA 76(3):1274–1278, 1979
Guidry C, Grinnell F: Studies on the mechanism of hydrated collagen gel reorganization by human skin fibroblasts. J Cell Sci 79:67–81, 1985
Harmatz P, Bloch K, Brown M, Walker W, Kleinman R: Intestinal adaptation during lactation in the mouse. I. Enhanced intestinal uptake of dietary protein antigen. Immunology 67:92–95, 1989
Israel E, Walker W: Host defense development in gut and related disorders. Pediatr Clin North Am 35:1–15, 1988
Barcellow-Hoff M, Aggeler H, Ram T, Bissell M: Functional differentiation and alveolar morphogenesis of primary mammary cultures on reconstituted basement membrane. Development 105:223–235, 1989
Schor A, Schor S, Allen T: Effects of culture conditions on the proliferation, morphology and migration of bovine aortic endothelial cells. J Cell Sci 62:267–285, 1983
Hall H, Farson D, Bissell M: Lumen formation by epithelial cell lines in response to collagen overlay: A morphogenetic model in culture. Proc Natl Acad Sci USA 79:4672–4676, 1982
Bissell DM, Arenson DM, Maher JJ, Roll FJ: Support of cultured hepatocytes by a laminin-rich gel. Evidence for a functionally significant subendothelial matrix in normal rat liver. J Clin Invest 79:801–812, 1987
Montgomery RK: Morphogenesisin vitro of dissociated fetal rat small intestinal cells upon an open surface and subsequent to collagen gel overlay. Dev Biol 117:64–70, 1986
Montgomery RK, Zinman HM, Smith BT: Organotypic differentiation of trypsin-dissociated fetal rat intestine. Dev Biol 100:181–189, 1983
Li ML, Aggeler J, Farson DA, Hatier C, Hassell J, Bissell MJ: Influence of a reconstituted basement membrane and its components on casein gene expression and secretion in mouse mammary epithelial cells. Proc Natl Acad Sci USA 84:136–140, 1987
Schafer I, Shapiro A, Kovach M, Lang C, Fratianne R: The interaction of human papillary and reticular fibroblasts and human keratinocytes in the contraction of three-dimensional floating collagen lattices. Exp Cell Res 183:112–125, 1989
Grinnell F: The activated keratinocyte: Up regulation of cell adhesion and migration during wound healing. J Trauma 30:S144-S149, 1990
Steinberg BM, Smith K, Colozzo M, Pollack R: Establishment and transformation diminish the ability of fibroblasts to contract a native collagen gel. J Cell Biol 87:304–308, 1980
Quaroni A, Isselbacher KJ, Ruoslahti E: Fibronectin synthesis by epithelial crypt cells of rat small intestine. Proc Natl Acad Sci USA 75(11):5548–5552, 1978
Nusrat A, Madara J: Characterization of intestinal epithelial cell motility in a model of restitution. Gastroenterology 100:A699, 1991
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Olson, A.D. Contraction of collagen gels by intestinal epithelial cells depends on microfilament function. Digest Dis Sci 38, 388–395 (1993). https://doi.org/10.1007/BF01316489
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01316489