Summary
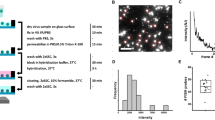
Current methods of detecting a human rhinovirus (HRV) infection are either based on isolation of virus in appropriate susceptible cell lines, which is time-consuming and requires considerable expertise, or are dependent on knowing the serotype. The extistence of over 100 immunologically distinct serotypes makes serotype specific assays, such as ELISA, unsuitable for general diagnostic assays. In this study a general rhinovirus assay is described which utilises synthetic oligonucleotides as probes in a filter hybridization assay. The probes are designed to bind to short but highly conserved regions of the rhinovirus genome. Indeed, the probes successfully detected all 57 rhinovirus serotypes tested. Furthermore, the test was used to demonstrate rhinovirus infection in clinical samples from 57 volunteers, inoculated with HRV, collected on six consecutive days. Clinical samples were taken prior to inoculation and on days 2–7 after inoculation. The filter hybridization assay gave results comparable to virus culture on days 2 and 3 post-inoculation, but was more sensitive on subsequent days.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Al-Nakib W, Higgins PG, Barrow GI, Tyrrell DAJ, Andries K, Vanden Bussche G, Taylor N, Janssen PAJ (1989) The suppression of colds in human volunteers challenged with rhinovirus by a new synthetic drug (R61837). Antimicrob Agents Chemother, in press
Craighead JE, Meier M, Cooley MH (1969) Pulmonary infection due to rhinovirus type 13. N Engl J Med 281: 1403–1404
Dearden C, Al-Nakib W (1987) Direct detection of rhinoviruses by an enzyme-linked immunosorbent assay. J Med Virol 23: 179–189
Duechler M, Skern T, Sommergruber W, Neubauer C, Gründler P, Fogy I, Blaas D, Küchler E (1987) Evolutionary relationships within the human rhinovirus genus: comparison of serotypes 89, 2, 14. Proc Natl Acad Sci USA 84: 2605–2609
Fox JP, Cooney MK, Hall CE, Fox HM (1985) Rhinoviruses in Seattle families: 1975–1979. Am J Epidemiol 122: 830–846
Gwaltney JM (1975) Medical reviews: rhinoviruses. Yale J Biol Med 48: 17–45
Hughes PJ, North C, Jellis CH, Minor PD, Stanway G (1983) The nucleotide sequence of human rhinovirus 1B: molecular relationships within the rhinovirus genus. J Gen Virol 69: 49–58
Hughes PJ, Phillips A, Minor PD, Stanway G (1987) The sequence of Coxsackievirus A21 polymerase gene indicates a remarkably close relationship to the polioviruses. Arch Virol 94: 141–147
Leckie GW (1988) Cloning and sequencing of the genome of human rhinovirus 9. PhD Thesis, University of Leicester, Leicester, England
Skern T, Sommergruber W, Blaas D, Gründler P, Fraundorfer F, Pieler C, Fogy I, Kuechler E (1985) Human rhinovirus 2: complete nucleotide sequence and proteolytic processing signals in the capsid protein region. Nucleic Acids Res 13: 2111–2126
Stanway G, Hughes PJ, Mountford RC, Minor PD, Almond JW (1984) The complete nucleotide sequence of a common cold virus: human rhinovirus 14. Nucleic Acid Res 12: 7859–7875
Stott EJ, Tyrrell DAJ (1968) Some improved techniques for the study of rhinoviruses using HeLa cells. Arch Ges Virusforsch 23: 236–244
Suggs SV, Hirose T, Miyake T, Kawashima EH, Johnson MJ, Kakura K, Wallace RB (1981) Use of synthetic oligodeoxyribonucleotides for the isolation of specific cloned DNA sequences. In: Brown DD, Fox CF (eds) Developmental biology using purified genes. Academic Press, New York, pp 683–697
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bruce, C.B., Al-Nakib, W., Almond, J.W. et al. Use of synthetic oligonucleotide probes to detect rhinovirus RNA. Archives of Virology 105, 179–187 (1989). https://doi.org/10.1007/BF01311355
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01311355