Summary
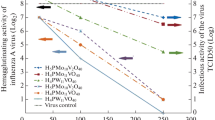
Human cytomegalovirus (HCMV), oxidized by sodium periodate (NaIO4), is incapable of giving rise to viral progeny in cell culture. At a NaIO4 concentration as low as 5 MM, there is a loss of at least 6 logs of viral infectivity which occurs very rapidly (less than 5 min). Further, the inactivation is a first-order reaction depending on the periodate concentration. Adsorption to the cell surface, penetration into cells, and penetration of the viral DNA into cell nuclei were found to occur identically in mock oxidized and oxidized HCMV. Since the carbohydrate moiety of viral glycoproteins was the target of periodate attack, these observations strongly suggest that the structural integrity of the sugar residues is not a perequisite for adsorption and penetration. Nevertheless, no evidence for viral DNA or protein synthesis was detected in cells infected with oxidized virus, and even after 3 weeks in culture, no cytopathic effect was observed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bachrach U, Reches B (1966) Enzymic assay for spermine and spermidine. Anal Biochem 17: 38–48
Bahnemann HG (1990) Inactivation of viral antigens for vaccine preparation with particular reference to the application of binary ethylenimine. Vaccine 8: 299–303
Balfour HHJ (1990) Management of cytomegalovirus disease with antiviral drugs. Rev Infect Dis 12: S849-S860
Beck E, Strohmaier K (1987) Subtyping of european foot-and-mouth disease strains by nucleotide sequence determination. J Virol 61: 1621–1629
Bowden RA, Meyers JD (1990) Prophylaxis of cytomegalovirus infection. Semin Hematol 27: 17–21
Dreesman GR, Suriano JR (1976) Alteration of adenovirus antigenic sites and infectivity by periodate oxidation. Virology 69: 700–709
Dunn S (1986) Effects of the modification of transfer buffer composition and the renaturation of proteins in gels on the recognition of proteins on western blots by monoclonal antibodies. Anal Biochem 157: 144–153
Elek SD, Stern H (1974) Development of a vaccine against mental retardation caused by cytomegalovirus infection in utero. Lancet 1: 1–5
Emanuel D (1990) Treatment of cytomegalovirus disease. Semin Hematol 27: 22–27
Gard S, Lycke E (1957) Inactivation of poliovirus by formaldehyde. Analysis of inactivation curves. Arch Ges Virusforsch 7: 471–482
Gehrz RC (1991) Human cytomegalovirus: biology and clinical perspectives. Adv Pediatr 38: 203–232
Gonczol E, Plotkin S (1990) Progress in vaccine development for prevention of human cytomegalovirus infection. Curr Top Microbiol Immunol 154: 255–274
Graves JH (1963) Formaldehyde inactivation of foot-and-mouth disease virus as applied to vaccine preparation. Am J Vet Res 24: 1131–1135
Ho M (1990) Epidemiology of cytomegalovirus infections. Rev Infect Dis 12: S701-S710
Jackson DA, Cook PR (1985) A general method for preparing chromatin containing intact DNA. EMBO J 4: 913–918
Jacobs JP, Jones CM, Baille JP (1970) Characteristics of a human diploïd cell designated MRC-5. Nature 227: 168–170
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Nowlin DM, Cooper NR, Compton T (1991) Expression of a human cytomegalovirus receptor correlates with infectibility of cells. J Virol 65: 3114–3121
Ogier G, Michal Y, Thomas V, Quash G, Rodwell JD (1994) Inhibition of type 5 advenovirus infectivity by periodate oxidation. Arch Virol 135: 43–60
Plotkin SA, Furukawa T, Zygraich N, Huygelen C (1975) Candidate cytomegalovirus strain for human vaccination. Infect Immun 12: 521–527
Plotkin SA, Starr SE, Friedman HM, Gonczol E, Brayman K (1990) Vaccines for the prevention of human cytomegalovirus infection. Rev Infect Dis 12: S827-S838
Porath A, McNutt RA, Smiley LM, Weigle KA (1990) Effectiveness and cost benefit of a proposed live cytomegalovirus vaccine in the prevention of congenital disease. Rev Infect Dis 12: 31–40
Quash G, Roch AM, Niveleau A, Grange J, Keolouangkhot T, Huppert J (1978) The preparation of latex particles with covalently bound polyamines, IgG and measles agglutinins and their use in visual agglutination tests. J Immunol Methods 22: 165–174
Reed LJ, Muench H (1938) A simple method of estimating fifty percent end points. Am J Hyg 27: 493–497
Snydman DR (1990) Cytomegalovirus immunoglobulins in the prevention and treatment of cytomegalovirus disease. Rev Infect Dis 12: S839-S848
Starr SE, Friedman HM, Plotkin SA (1990) The status of cytomegalovirus vaccine. Rev Infect Dis 13: S964-S965
Taylor HP, Cooper NR (1990) The human cytomegalovirus receptor on fibroblasts is a 30-kilodalton membrane protein. J Virol 64: 2484–2490
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76: 4350–4354
Wesslén T, Dinter Z (1957) The inactivation of foot-and-mouth disease virus by formalin. Arch Ges Virusforsch 7: 394–402
Wesslén T, Lycke E, Gard S, Olin G (1957) Inactivation of poliomyelitis virus by formaldehyde. Arch Ges Virusforsch 7: 125–135
Zaia AJ (1990) Epidemiology and pathogenesis of cytomegalovirus diseases. Semin Hematol 27: 5–10
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Geoffroy, F., Ogier, G., Chantepie, J. et al. Inactivation of human cytomegalovirus by sodium periodate oxidation. Archives of Virology 135, 61–74 (1994). https://doi.org/10.1007/BF01309765
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01309765