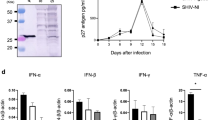
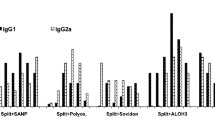
Summary
Oral and/or intranasal inoculation of susceptible mouse genotypes with the JHM strain of mouse hepatitis virus (MHV-JHM) consistently results in T cell dysfunction as reflected by in vitro proliferative responses to mitogens or allogeneic cells. One approach to examining the mechanism responsible for the observed functional T cell suppression is to determine whether virus replication is required for its induction. To this end, mice were inoculated oronasally with MHV-JHM that was inactivated with short-wave ultraviolet light, betapropiolactone or psoralen. Mice were also inoculated with live MHV-JHM after recovery from homotypic or heterotypic MHV infection. Spleen cells from BALB mice inoculated oronasally with inactivated MHV-JHM yielded extremely variable in vitro proliferative responses after concanavalin A stimulation. MHV-susceptible mice exposed oronasally or intraperitoneally to virus inactivated by any of the minimum effective treatments failed to seroconvert. Immunization with psoralen-treated virus intraperitoneally in Freund's complete adjuvant or oronasally failed to protect from live virus challenge, but survivors had elevated virus-specific serum IgG antibody titers compared to mock-immunized controls at two weeks post-challenge. Spleen cells from mice that were challenged after recovery from homotypic live virus infection did not exhibit the profound in vitro T cell suppression normally observed during the acute stage of primary infection. In contrast, MHV-JHM challenge of mice vaccinated with heterotypic live MHV-S resulted in significantly depressed in vitro T cell function. The combined data suggest that either virus replication or exposure to more concentrated antigen may be required for induction of the dramatic T cell dysfunction that occurs as a consequence of MHV-JHM infection as well as for a detectable MHV-specific humoral response.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barthold SW, Smith AL (1987) Response of genetically susceptible and resistant mice to intranasal inoculation with mouse heptatitis virus JHM. Virus Res 7: 225–239
Barthold SW, Smith AL (1989) Virus strain specificity of challenge immunity to coronavirus. Arch Virol 104: 187–196
Bia FJ, Thornton GF, Main AJ, Fong CKY, Hsiung GD (1980) Western equine encephalitis mimicking herpes simplex encephalitis. J Am Med Assoc 244: 367–369
Boorman GA, Luster MI, Dean JH, Campbell ML, Lauer LA, Talley FA, Wilson RE, Collins MJ (1982) Peritoneal macrophage alterations caused by naturally occurring mouse hepatitis virus. Am J Pathol 106: 110–117
Braunsteiner H, Friend C (1954) Viral hepatitis associated with transplantable mouse leukemia. J Exp Med 100: 665–677
Brownstein DG, Barthold SW (1982) Mouse hepatitis virus immunofluorescence in formalin- or Bouin's-fixed tissues using trypsin digestion. Lab Anim Sci 32: 37–39
Buckley SM, Casals J (1970) Lassa fever, a new virus disease of man from West Africa. III. Isolation and characterization of the viurs. Am J Trop Med Hyg 19: 680–691
Carter JJ, Weinberg AD, Pollard A, Reeves R, Magnuson JA, Magnuson NS (1989) Inhibition of T-lymphocyte mitogenic responses and effects on cell functions by bovine herpesvirus 1. J Virol 63: 1525–1530
Casebolt DB, Spalding DM, Schoeb TR, Lindsey JR (1987) Suppression of immune response induction in Peyer's patch lymphoid cells from mice infected with mouse hepatitis virus. Cell Immunol 109: 97–103
Cimino GD, Gamper HB, Isaacs ST, Hearst JE (1985) Psoralens as photoactive probes of nucleic acid structure and function: organic chemistry, photochemistry, and biochemistry. Annu Rev Biochem 54: 1151–1193
Colonna Romano G, Dieli F, Abrignani S, Salerno A, Colizzi V (1986) Inhibition of lymphocyte mitogenesis in mice infected with Newcastle disease virus: viral interference with the interleukin system. Immunology 57: 373–378
Dempsey WL, Smith AL, Morahan PS (1986) Effect of inapparent murine hepatitis virus infections on macrophages and host resistance. J Leuk Biol 39: 559–565
de Souza MS, Smith AL (1991) Characterization of accessory cell function during acute infection of BALB/cByJ mice with mouse hepatitis virus (MHV), strain JHM. Lab Anim Sci 41: 112–118
de Souza MS, Smith AL, Bottomly K (1991) Infection of BALB/cByJ mice with the JHM strain of mouse hepatitis virus alters in vitro splenic T cell proliferation and cytokine production. Lab Anim Sci 41: 99–105
Engers HD, Louis JA, Zubler RH, Hirt B (1981) Inhibition of T cell-mediated functions by MVM(i), a parvovirus closely related to minute virus of mice. J Immunol 127: 2280–2285
Gillis S, Ferm MM, Ou W, Smith KA (1978) T cell growth factor: parameters of production and a quantitative microassay for activity. J Immunol 120: 2027–2032
Hanson CV (1983) Inactivation of viruses for use as vaccines and immunodiagnostic reagents. In: de la Maza LM, Peterson EM (eds) Medical virology II. Elsevier, New York, pp 45–79
Harty JT, Plagemann PG (1988) Formalin inactivation of the lactate dehydrogenase-elevating virus reveals a major neutralizing epitope not recognized during natural infection. J Virol 62: 3210–3216
Homberger FR, Smith AL, Barthold SW (1991) Detection of rodent coronaviruses in tissues and cell cultures using polymerase chain reaction. J Clin Microbiol 29: 2789–2793
Hu-Li J, Ohara J, Watson C, Tsang W, Paul WE (1989) Derivation of a T cell line that is highly responsive to IL-4 and IL-2 (CT.4R) and of an IL-2 hyporesponsive mutant of that line (CT.4S). J Immunol 142: 800–807
Kraft V, Meyer B (1986) Diagnosis of murine infections in relation to test methods employed. Lab Anim Sci 36: 271–276
Lindsey JR (1986) Prevalence of viral and mycoplasmal infections in laboratory rodents. In: Bhatt PN, Jacoby RO, Morse HC, New A (eds) Viral and mycoplasmal infections of laboratory rodents. Effects on biomedical research. Academic Press, New York, pp 801–808
LoGrippo GA, Hartman FW (1955) Antigenicity of beta-propiolactone-inactivated virus vaccines. J Immunol 75: 123–128
Lucas CJ, Ubels-Postma JC, Rezee A, Galama JMD (1978) Activation of measles virus from silently infected human lymphocytes. J Exp Med 148: 940–952
Lussier G, Descoteaux J-P (1986) Prevalence of natural virus infection in laboratory mice and rats used in Canada. Lab Anim Sci 36: 145–148
McChesney MB, Altmann A, Oldstone MBA (1988) Suppression of T lymphocyte function by measles virus is due to cell cycle arrest in G1. J Immunol 140: 1269–1273
Margolese RG, Israel E, Wainberg MA (1980) Non-specific inhibition by virus particles of human lymphocyte mitogenesis. Clin Exp Immunol 41: 243–251
Mosmann TR, Cherwinski H, Bond MW, Giedlin MA, Coffman RL (1986) Two types of murine T helper cell clones. I. Definition according to profile of lymphokine activities. J Immunol 136: 2348–2357
Ohara J, Paul WE (1985) B cell stimulatory factor BSF-1: production of a monoclonal antibody to and molecular characterization of B-cell stimulatory factor-1. Nature 315: 333–336
Small JD, New AE (1981) Prevention and control of mousepox. Lab Anim Sci 31: 616–621
Smith AL (1983) An immunofluorescence test for detection of serum antibody to rodent coronaviruses. Lab Anim Sci 33: 157–160
Smith AL, Barthold SW, de Souza MS, Bottomly K (1991) The role of gamma interferon in infection of susceptible mice with murine coronavirus, MHV-JHM. Arch Virol 121: 89–100
Smith AL, Bottomly K, Winograd DF (1987) Altered splenic T cell function of BALB/cByJ mice infected with mouse hepatitis virus or Sendai virus. J Immunol 138: 3426–3430
Smith AL, Casals J, Main AJ (1983) Antigenic characterization of Tettnang virus: Complications caused by passage of the virus in mice from a colony enzootically infected with mouse hepatitis virus. Am J Trop Med Hyg 32: 1172–1176
Smith AL, Winograd DF (1986) Two enzyme immunoassays for the detection of antibody to rodent coronaviruses. J Virol Methods 14: 335–343
Smith AL, Winograd DF, de Souza MS (1991) In vitro splenic T cell responses of diverse mouse genotypes after oronasal exposure to mouse hepatitis virus, strain JHM. Lab Anim Sci 41: 106–111
Strayer DS, Skaletsky E, Leibowitz JL (1986) Inhibition of virus replication does not alter malignant rabbit fibroma virus-induced immunosuppression. Clin Exp Immunol 66: 25–36
Suzuki E, Matsubara J, Saito M, Muto T, Nakagawa M, Imaizumi K (1982) Serological survey of laboratory rodents for infection with Sendai virus, mouse hepatitis virus, reovirus type 3 and mouse adenovirus. Jpn J Med Sci Biol 35: 249–254
Wainberg MA, Israel E (1980) Viral inhibition of lymhocyte mitogenesis. I. Evidence for nonspecificity of the effect. J Immunol 124: 64–70
Wainberg MA, Portnoy JD, Clecner B, Hubschman S, Lagace-Simard J, Rabinovitch N, Remer Z, Mendelson J (1985) Viral inhibition of lymphocyte proliferative responsiveness in patients suffering from recurrent lesions caused by herpes simplex virus. J Infect Dis 152: 441–448
Watson J, Gillis S, Marbrook J, Mochizuki D, Smith KA (1979) Biochemical and biological characterization of lymphocyte regulatory molecules. I. Purification of a class of murine lymphokines. J Exp Med 150: 849–861
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smith, A.L., de Souza, M.S., Finzi, D. et al. Responses of mice to murine coronavirus immunization. Archives of Virology 125, 39–52 (1992). https://doi.org/10.1007/BF01309627
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01309627