Summary
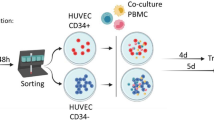
Human primary endothelial cell cultures, derived from umbilical vein (HUVEC), can be infected by different strains of HIV-1, but mature virus production remains undetectable both in supernatants and in cellular extracts. Yet viral DNA is transiently detectable during the first days of infection, but progressively declines during the subsequent days. This finding is characteristic of abortive infections. Co-culture of HUVEC carrying HIV DNA with activated peripheral blood mononuclear cells or with CD 4-positive lymphoid cells elicited a massive cpe (syncytia formation and cell degeneration) in the latter cells, caused by the establishment of productive HIV-1 infection. HUVEC infected in the presence of AZT were significantly impaired in the ability to transmit the infection of CD 4-positive cells, indicating that active DNA synthesis is required in HUVEC before rescue by CD 4-positive cells.
These results are of interest in view of the possibility that endothelial cells can play a role in the transmission of HIV-1 infection from infected pregnant women to the foetuses, and, more generally, suggest a potential role of endothelial cells as a transient reservoir of HIV-1.
Similar content being viewed by others
References
Harouse JM, Kunsch C, Hartle HT, Laughlin MA, Hoxie JA, Wigdahl B, Gonzalez-Scarano F (1989) CD 4-independent infection of human neural cells by human immunodeficiency virus type 1. J Virol 63: 2527–2533
Kunsch C, Hartle HT, Wigdahl B (1989) Infection of fetal dorsal root ganglion glial cells with human immunodeficiency virus type 1 involves an entry mechanism independent of the CD 4 T4A epitope. J Virol 63: 5054–5061
Clapham PR, Weber JN, Whitby D, McIntosh K, Dalgleish A, Maddon PJ, Deen KC, Sweet RW, Weiss RA (1989) Soluble CD 4 blocks the infectivity of diverse strains of HIV and SIV for T cells and monocytes but not for brain and muscle cells. Nature 337: 368–373
Cao Y, Friedman-Kien AE, Huang Y, Li XL, Mirabile M, Moudgil T, Zucker-Franklin D, Ho DD (1990) CD 4-independent, productive human immunodeficiency virus type 1 infection of hepatoma cell lines in vitro. J Virol 64: 2553–2559
Tateno M, Gonzales-Scarano F, Levy JA (1989) Human immunodeficiency virus can infect CD 4-negative human fibroblastoid cells. Proc Natl Acad Sci USA 86: 4287–4290
Dolei A, Serra C, Arca MV, Toniolo A (1992) Acute HIV-1 infection of CD 4+ human lung fibroblast. AIDS 6: 232–234
Fantini J, Yahi N, Baghdiguian S, Chermann JC (1992) Human colon epithelial cell productively infected with human immunodeficiency virus show impaired differentiation and altered secretion. J Virol 66: 580–585
Re MC, Furlini G, Cenacchi G, Preda P, La Placa M (1991) Human immunodeficiency virus type 1 infection of endothelial cells in vitro. Microbiology 14: 149–152
Steffan AM, Lafon ME, Gendrault JL, Schweitzer C, Royer C, Jaeck D, Arnaud JP, Schmitt MP, Aubertin AM, Kirn A (1992) Primary cultures of endothelial cells from the human liver sinusoid are permissive for human immunodeficiency virus type 1. Proc Natl Acad Sci USA 89: 1582–1586
Perez LG, O'Donnell MA, Stephens EB (1992) The transmembrane glycoprotein of human immunodeficiency virus type 1 induces syncytium formation in the absence of the receptor binding glycoprotein. J Virol 66: 4134–4143
Beilke MA (1989) Vascular endothelium in immunology and infectious disease. Rev Infect Dis 41: 273–283
Pober JS, Ramzi RS (1990) The role of endothelial cells in inflammation. Transplantation 50: 537–544
Pober JS, Doukas J, Hughes CC, Savage CO, Munro JM, Cotran RS (1990) The potential roles of vascular endothelium in immune reactions. Hum Immunol 28: 258–262
Hirschberg H (1981) Presentation of viral antigens by human vascular endothelial cells in vitro. Hum Immunol 2: 235–246
Miossec P, Ziff M (1986) Immune interferon enhances the production of interleukin-1 by human endothelial cells stimulated with lipopolysaccharide. J Immunol 137: 2848–2852
Shalaby MR, Waage A, Espevik T (1989) Cytokine regulation of interleukin-6 production by human endothelial cells. Cell Immunol 121: 372–382
Teitel JM, Shore A, McBarron J, Schiavone A (1989) Enhanced T cell activation due to combined stimulation by both endothelial cells and monocytes. Scand J Immunol 29: 165–173
Baroni CD, Pezzella F, Mirolo M, Ruco LP, Rossi GB (1986) Immunohistochemical demonstration of p 24 HTLV III major core protein in different cell types within lymph nodes from patients with lymphadenopathy syndrome (LAS). Histopathology 10: 5–13
Pomerantz RJ, De la Monte SM, Donegan P, Rota TR, Vogt MW, Craven DE, Hirsch MS (1988) Human immunodeficiency virus (HIV) infection of the uterine cervix. Ann Intern Med 108: 321–327
Wiley CA, Schrier RD, Nelson JA, Lampert PW, Oldstone MBA (1986) Cellular localization of human immunodeficiency virus infection within the brains of acquired immune deficiency syndrome patients. Proc Natl Acad Sci USA 83: 7089–7093
Ades EW, Comans TW, Nicholson JKA, Browning SW (1993) Lack of evidence that human immunodeficiency virus can infect human endothelial cells in vitro. J Acquir Immune Defic Syndr 6: 104–105
Lafon ME, Steffan AM, Gendrault JL, Klein-Soyer C, Gloeckler-Tondre L, Royer C, Kirn A (1992) Interaction of human immunodeficiency virus with human macrovascular endothelial cells in vitro. AIDS Res Hum Retroviruses 8: 1567–1570
Cenacchi G, Re MC, Furlini G, La Placa M, Binetti G, Rapezzi C, Alampi G, Magnani B (1990) Human immunodeficiency virus type 1 antigen detection in endomyocardial biopsy: an immunomorphological study. Microbiology 13: 145–149
Jaffe EA (1980) Culture of human endothelial cells derived from cord veins. Transplant Res 12: 49–62
Maciag T, Hoover GA, Stemerman MB, Weinstein R (1981) Serial propagation of human endothelial cells in vitro. J Cell Biol 91: 420
Shimizu Y, Newman W, Tanaka Y, Shaw S (1992) Lymphocyte interactions with endothelial cells. Rev Immunol Today 13: 106–112
Dianzani F, Antonelli G, Capobianchi MR, De Marco F (1988) Replication of human immunodeficiency virus: yield of infectious virus under single growth cycle conditions. Arch Virol 103: 127–131
Popovic M, Sarngsharau MG, Read E, Gallo RC (1984) Detection, isolation, and continuous production of cytopathic retroviruses (HTLV-III) from patients with AIDS and pre-AIDS. Science 224: 497–500
Tang SB, Levy JA (1991) Inactivation of HIV-1 by trypsin and its use of demonstrating specific virus infection of cells. J Virol Methods 33: 39–46
Dianzani F, Antonelli G, Turriziani O, Guanmu D, Capobianchi MR, Riva E (1992) In vitro selection of human immunodeficiency virus type 1 resistant to 3′-azido-3′-deoxythymidine. Antiviral Res 18: 39–52
Dianzani F, Capobianchi MR, Antonelli G, Amicucci P, De Marco F (1989) Susceptibility of human immunodeficiency virus to antiviral agents measured by infectious virus yield reduction. Antiviral Res 11: 299–306
Chiodi F, Fuerstenberg S, Gidlund M, Asjo B, Fenyo EM (1987) Infection of brain-derived cells with the human immunodeficiency virus. J Virol 61: 1244–1247
Cheng-Mayer C, Rutka JT, Rosenblum ML, McHugh T, Stites DP, Levy JA (1987) Human immunodeficiency virus can productively infect cultured human glial cells. Neurobiology 84: 3526–3530
Kunsch C, Wigdahl B (1989) Transient expression of human immunodeficiency virus type 1 genome results in a non-productive infection in human fetal dorsal root ganglia glial cells. Virology 173: 715–722
Cameron PU, Freudenthal PS, Barker JM, Gezelter S, Inaba K, Steinman RM (1992) Dendritic cells exposed to human immunodeficiency virus type 1 transmit a vigorous cytopathic infection to CD 4+ T cells. Science 257: 383–387
Sattentau QJ, Dalgleish AG, Weiss RA, Beverley PC (1986) Epitopes of the CD 4 antigen and HIV infection. Science 234: 1120–1123
Bhat S, Spitalnik SL, Gonzalez-Scarano F, Silberberg DH (1991) Galactosyl ceramide or a derivative is an essential component of the neural receptor for human immunodeficiency virus type 1 envelope glycoprotein. Proc Natl Acad Sci USA 88: 7131–7134
Yahi N, Baghdiguian S, Moreau H, Fantini J (1992) Galactosyl ceramide (or a closely related molecule) is the receptor for human immunodeficiency virus type 1 on human colon epithelial HT 29 cells. J Virol 66: 4848–4854
Zack JA, Haislip AM, Krogstad P, Chen ISY (1992) Incompletely reverse-transcribed human immunodeficiency virus type 1 genomes in quiescent cells can function as intermediates in the retroviral life cycle. J Virol 66: 1717–1725
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Scheglovitova, O., Capobianchi, M.R., Antonelli, G. et al. CD 4-positive lymphoid cells rescue HIV-1 replication from abortively infected human primary endothelial cells. Archives of Virology 132, 267–280 (1993). https://doi.org/10.1007/BF01309538
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01309538