Summary
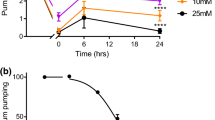
Feeding behavior ofNoctiluca miliaris cultured together with a food organism,Dunaliella tertiolecta, an unicellular green alga, was studied. The feeding process was separated into successive four steps. (1) Food is trapped on the tentacle tip covered with mucus secreted from the alveolar pore. (2) The food is carried by bending the tentacle to the cytostome which is in the deepest part of the oral pouch. (3) Particular, vigorous cytoplasmic aggregation occurs toward the cytostome where the membrane of the cytoplasmic mass is exposed to external medium. (4) The membrane invaginates at the cytostome and the food is ingested in the cytoplasm. The cytoplasmic aggregation is applicable as an indicator of the experimental induction of feeding behavior. Extract of the food, amino acids or glutathione added into the bath did not effectively induce the feeding. Mechanical stimulus applied to around the cytostome induced feeding only in cells of a certain age. Lowering SO4 2− from 30 mM to 2 mM in the bath always induced feeding within a few minutes. Ionic factors of 1 mM Ca2−, 20 mM Mg2+, and 400 mM Cl− or more in the bath were essential for the induction of feeding. Application of an inward electric current (bath to vacuole, 25 nA or more) also induced the same response as that obtained by lowering SO4 2− concentration.
Similar content being viewed by others
References
Cabantchik, Z. I., Knauf, P. A., Rothstein, A., 1978: The anion transport system of the red blood cell. The role of membrane protein evaluated by the use of probes. Biochim. biophys. Acta515, 239–302.
Dodge, J. D., Crawford, R. M., 1970: A survey of thecal fine structure in theDinophyceae. Bot. J. Linn. Soc.63, 53–67.
Eckert, R., Sibaoka, T., 1967: Bioelectric regulation of tentacle movement in a dinoflagellate. J. exp. Biol.47, 433–446.
Garrels, R. M., Thompson, M. E., 1962: A chemical model for sea water at 25°C and one atmosphere total pressure. Amer. J. Sci.260, 57–66.
Jahn, T. L., Bovee, E. C., 1969: Protoplasmic movements within cells. Physiol. Rev.49, 793–862.
Kofoid, C. A., Swezy, O., 1921: The free-living unarmoredDinoflagellata. Mem. Univ. Calif.5, 1–562.
Komnick, H., Stockem, W., Wohlfarth-Bottermann, K. E., 1973: Cell motility: Mechanisms in protoplasmic streaming and ameboid movement. Int. Rev. Cytol.34, 169–249.
Narahashi, T., 1974: Chemicals as tools in the study of excitable membranes. Physiol. Rev.54, 813–889.
Nawata, T., Sibaoka, T., 1976: Ionic composition and pH of the vacuolar sap in marine dinoflagellateNoctiluca. Plant Cell Physiol.17, 265–272.
— —, 1979: Coupling between action potential and bioluminescence inNoctiluca. Effects of inorganic ions and pH in vacuolar sap. J. comp. Physiol.134, 137–149.
Pratje, A., 1921:Noctiluca miliaris Suriray. Beiträge zur Morphologie, Physiologie und Cytologie.I. Morphologie und Physiologie. Arch. Protistenk.42, 1–98.
Soyer, M. O., 1970: Les ultrastructures liées aux fonctions de relation chezNoctiluca miliaris S. (Dinoflagellata). Z. Zeilforsch.104, 29–55.
Spurr, A. R., 1969: A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res.26, 31–43.
Taylor, D. L., Condeelis, J. S., 1979: Cytoplasmic structure and contractility in amoeboid cells. Int. Rev. Cytol.56, 57–144.
Wolf, K. V., Hoffmann, H. U., Stockem, W., 1981: Studies on microplasmodia ofPhysarum polycephalum: II. Fine structure and function of the mucous layer. Protoplasma107, 345–359.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nawata, T., Sibaoka, T. Experimental induction of feeding behavior inNoctiluca miliaris . Protoplasma 115, 34–42 (1983). https://doi.org/10.1007/BF01293578
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01293578