Summary
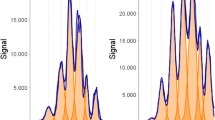
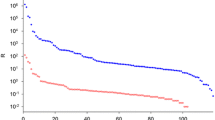
We have developed a new procedure, including three affinity chromatography steps, micro-reversed phase high pressure liquid chromatography (mR-HPLC) and Western blotting/mass spectrometric analysis to study central nervous system (CNS) specific proteins in human cerebrospinal fluid (CSF) in order to find biochemical markers for neuronal and synaptic function and pathology in degenerative brain disorders. After the three affinity chromatography steps, intended to remove interfering serum proteins from CSF, mR-HPLC revealed four major peaks, which by both Western blotting and mass spectrometric analyses were found to correspond to β2-microglobulin, cystatin C, transthyretin (TTR) and asialotransferrin. When comparing these peaks in CSF from Alzheimer's disease (AD) patients and age-matched healthy controls, a reduction of the brain-specific TTR was found. Therefore we quantified TTR in CSF and serum samples from 8 patients with early onset AD (EAD), 18 patients with late onset AD (LAD), 8 patients with vascular dementia (VAD) and 18 healthy individuals using a nephelometric method. CSF-TTR was divided into barrier-dependent and barrier-independent TTR. The barrier-independent i.e. brain-specific TTR was significantly reduced in the EAD group compared to the controls. Transthyretin has been found to be present in the senile plaques in AD, and to specifically bind to β/A4 protein, the major component of the amyloid deposits in AD. Therefore, the reduction of the transthyretin-isoform in CSF in AD may reflect an absorption of transthyretin to the amyloid deposits in the senile plaques.
Similar content being viewed by others
References
Åkerström B, Björk L (1986) A physiochemical study of Protein G, a molecule with unique immunoglobulin G binding properties. J Biol Chem 261: 10240–10247
Coleman PP, Flood DG (1987) Neuron number and dendrite extent in normal ageing and Alzheimer's disease. Neurobiol Aging 78: 151–164
Felding P (1984) Prealbumin, metabolic and chemical studies. Thesis, University of Malmö
Felgenhauer K (1974) Protein size and cerebrospinal fluid composition. Klin Wochenschr 52: 1158–1164
Folstein M, Folstein S, McHugh P (1975) “Mini-Mental State”: a practical method for grading the cognitive state of patients for the clinician. J Psychiat Res 12: 189–198
Golabek A, Marques MA, Lalowski M, Wisniewski T (1995) Amyloid beta binding protein in vitro and in normal human cerebrospinal fluid. Neurosci Lett 191 (1–2): 79–82
Harrington MG, Merril CR, Torrey EF (1985) Differences in cerebrospinal fluid between patients with schizophrenia and normal persons. Clin Chem 31 (5): 722–726
Kanda Y, Goodman DWS, Canfield RE, Morgan FJ (1974) The amino acid sequence of human plasma prealbumin. J Biol Chem 249: 6796–6805
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage. Nature 227: 680–685
Leary JJ, Brigati DJ, Ward DC (1983) Rapid and sensitive colorimetric method for visualising biotin-labelled DNA probes hybridised to DNA or RNA immobilised in nitro-cellulose: bio-blots. Proc Natl Sci USA 80: 4045–4049
Leone MG, Saso L, Del Vecchio A, Mo M, Silverstrini B, Cleng CY (1993) Micropurification of two human cerebrospinal fluid proteins by high performance electrophoresis chromatography. J Neurochem 61: 533–540
Mattila KM, Frey H (1994) Alzheimer brain proteins investigated by two-dimensional gel electrophoresis with immobilised pH gradients in the first dimension. Electrophoresis 15(5): 721–725
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work group under auspices of department of health and human services task force on Alzheimer's disease. Neurology 34: 939–944
Näslund J, Schierhorn A, Hellman U, Lannfelt L, Roses AD, Tjerrberg LO, Silberring J (1994) Relative abundance of Alzheimer A beta amyloid peptide variants in Alzheimer's disease and normal ageing. Proc Natl Acad Sci USA 30: 91(18) 8378–8382
PhastSystemTM Separation Technique File No 100. Pharmacia, Uppsala, Sweden
PhastSystemTM Development Technique File No 210. Pharmacia, Uppsala, Sweden
Riisoen H (1988) Reduced prealbumin (transthyretin) in CSF of severely demented patients with Alzheimer's disease. Acta Neurol Scand 78: 455–459
Schwartzman A, Gregori L, Vitek MP, Lyubski S, Strittmatter WJ, Enghilde JJ, Bhasini R, Silverman J, Weisgraber KH, Coyle PK, Zagorski MG, Talafous J, Eisenberg M, Saunders AM, Roses AD, Goldgaber D (1994) Transthyretin sequesters amyloid β protein and prevents amyloid formation. Proc Natl Acad Sci USA 91: 8168–8372
Selkoe DJ (1989) Ageing, amyloid and Alzheimer's disease. N Engl J Med 320: 1484–1487
Shirahama T, Skinner M, Westermark P, Rubinow A, Cohen AS, Brun A, Kemper TL (1982) Senile cerebral amyloid; prealbumin as a common constituent in the neurite plaque, in the neurofibrillary tangle and in the microangiopathic lesion. Am J Pathol 107: 41–50
Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R, Hansen LA, Katzman R (1991) Physical basis of cognitive alterations in Alzheimer's disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol 30: 572–580
Tomlinson BE, Corsellis JAN (1984) Ageing and the dementias. In: Hume Adams J, Corsellis JAN, Duchen LW (eds) Greenfield's neuropathology. Edward Arnold, London, pp 951–1025
Thompson EJ (1988) The CSF proteins: a biomedical approach. Elsevier, Amsterdam, pp 67–85
Travis J, Pahnell R (1973) Selective removal of albumin from plasma by affinity chromatography. Clin Chem Acta 49: 49–52
Wikkelsö C, Blomstrand C, Rönnbäck L (1980) Separation of specific proteins-a methodological study. J Neurol Sci 44: 247–257
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Davidsson, P., Ekman, R. & Blennow, K. A new procedure for detecting brain-specific proteins in cerebrospinal fluid. J. Neural Transmission 104, 711–720 (1997). https://doi.org/10.1007/BF01291888
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01291888