Summary
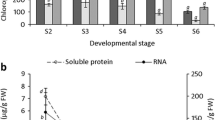
Previously, we showed that all greening mesophyll cells in the coleoptiles of rice (Oryza sauva L. cv. Nippon-bare) follow the identical program of senescence, which features the early degradation of chloroplast DNA (cpDNA) and subsequent nuclear condensation and disorganization. Following the coleoptile study, we analyzed the senescence-associated changes in the blade of the second leaf of rice at the tissue and cellular levels. Under the experimental conditions, the second leaf started to elongate rapidly 2 days after sowing and emerged on day 3. The blade of the second leaf completed its growth on day 4, although the sheath continued to grow until day 7. The amount of soluble protein and chlorophyll (Chl) per blade reached a maximum on day 7, and then declined. When blades were divided into three parts (the tip, mid-region, and base), levels of both soluble protein and Chl in the tip segment peaked earlier and decreased at a faster rate than in the other parts, demonstrating a longitudinal gradient of senescence from the tip to the base of the blade. In cross sections through the center of the tip and base segments, all the mesophyll cells senesced synchronously. They passed through the following steps in order: (i) degradation of cpDNA, (ii) decrease in the size of the chloroplast with degeneration of the chloroplast inner membranes, and (iii) condensation and disorganization of the nuclei. Although some differences were shown between the coleoptile and the second leaf in the timing and rate of each event, the order of those senescence-related events was conserved, suggesting an identical program of senescence exists in rice leaves.
Similar content being viewed by others
Abbreviations
- Chl:
-
chlorophyll
- cpDNA:
-
chloroplast DNA
- cpnucleoid:
-
chloroplast nucleoid
- DAPI:
-
4′,6-diamidino-2-phenylindole
- DiOC7 :
-
3,3′-dihexyloxacarbocyanine iodide
- VB:
-
vascular bundle
- VIMPCS:
-
video-intensified microscope photon-counting system
References
Buchanan-Wollaston V (1997) The molecular biology of leaf senescence. J Exp Bot 48: 181–199
Campbell R, Drew MC (1983) Electron microscopy of gas space (aerenchyma) formation in adventitious roots ofZea mays L. subjected to oxygen shortage. Planta 157: 350–357
Ford DM, Shibles R (1988) Photosynthesis and other traits in relation to chloroplast number during soybean leaf senescence. Plant Physiol 86: 108–111
Fujie M, Kuroiwa H, Kawano S, Mutoh S, Kuroiwa T (1994) Behavior of organelles and their nucleoids in the shoot apical meristem during leaf development inArabidopsis thaliana L. Planta 194: 395–405
Hashimoto H, Kura-Hotta M, Katoh S (1989) Changes in protein content and in the structure and number of chloroplasts during leaf senescence in rice seedlings. Plant Cell Physiol 30: 707–715
He CJ, Morgan PW, Drew MC (1996) Transduction of an ethylene signal is required for cell death and lysis in the root cortex of maize during aerenchyma formation induced by hypoxia. Plant Physiol 112: 463–472
Horton RF, Osborne DJ (1967) Senescence, abscission and cellulase activity inPhaseolus vulgaris. Nature 214: 1086–1088
Hoshikawa K (1989) The growing rice plant: an anatomical monograph. Nobunkyo, Tokyo
Inada N, Sakai A, Kuroiwa H, Kuroiwa T (1998a) Three-dimensional analysis of the senescence program in rice (Oryza sauva L.) coleoptiles: investigations of tissues and cells by fluorescence microscopy. Planta 205: 153–164
— — — — (1998b) Three-dimensional analysis of the senescence program in rice (Oryza saliva L.) coleoptiles: investigations by fluorescence microscopy and electron microscopy. Planta 206: 585–597
Jacobson MD, Weil M, Raff MC (1997) Programmed cell death in animal development. Cell 88: 347–354
Kerr JFR, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26: 239–257
Kura-Hotta M, Hashimoto H, Satoh K, Katoh S (1990) Quantitative determination of changes in the number and size of chloroplasts in naturally senescing leaves of rice seedlings. Plant Cell Physiol 31: 33–38
Kuroiwa T, Suzuki T (1980) An improved method for the demonstration of the in situ chloroplast nuclei in higher plants. Cell Struct Funct 5: 195–197
—, Miyamura S, Kawano S, Hizume M, Toh-e A, Miyakawa I, Sando N (1986) Cytological characterization of NOR in the bivalent ofSaccharomyces cerevisiae. Exp Cell Res 165: 199–206
—, Fujie M, Mita T, Kuroiwa H (1991) Application of embedding of samples in Technovit 7100 resin to observations of small amounts of DNA in the cellular organelles associated with cytoplasmic inheritance. Appl Fluoresc Tech 3: 23–25
Lamppa GK, Elliot LV, Bendich AJ (1980) Changes in chloroplast number during pea leaf development: an analysis of a protoplast population. Planta 148: 437–443
Lawrence ME, Possingham JV (1986) Microspectrofluorometric measurement of chloroplast DNA in dividing and expanding leaf cells ofSpinacia oleracea. Plant Physiol 81: 708–710
Martinoia E, Heck U, Dalling MJ, Matile PH (1983) Changes in chloroplast number and chloroplast constituents in senescing barley leaves. Biochem Physiol Pflanzen 178: 147–155
Matile PH (1973) Cell wall degradation in senescing tobacco leaf discs. Experimentia 30: 98–99
—, Hörtensteiner S, Thomas H, Kräutler B (1996) Chlorophyll breakdown in senescent leaves. Plant Physiol 112: 1403–1409
Noodén LD, Leopold AC (1988) Senescence and aging in plants. Academic Press, California
—, Guiamét JJ, John I (1997) Senescence mechanisms. Physiol Plant 101: 746–753
Ono K, Hashimoto H, Katoh S (1995) Changes in the number and size of chloroplasts during senescence of primary leaves of wheat grown under different conditions. Plant Cell Physiol 36: 9–17
Pennell RI, Lamb C (1997) Programmed cell death in plants. Plant Cell 9: 1157–1168
Scott NS, Possingham JV (1980) Chloroplast DNA in expanding spinach leaves. J Exp Bot 31: 1081–1092
Smart CM (1994) Gene expression during leaf senescence. New Phytol 126: 419–448
Sodmergen, Kawano S, Tano S, Kuroiwa T (1989) Preferential digestion of chloroplast nuclei (nucleoids) during senescence of the coleoptile ofOryza sativa. Protoplasma 152: 65–68
— — — — (1991) Degradation of chloroplast DNA in second leaves of rice (Oryza sativa) before leaf yellowing. Protoplasma 160: 89–98
Wardley TM, Bhalla PL, Dalling MJ (1984) Changes in the number and composition of chloroplasts during senescence of mesophyll cells of attached and detached primary leaves of wheat (Triticum aestivum L.). Plant Physiol 75: 421–424
Webb J, Jackson MB (1986) A transmission and cryo-scanning electron microscopy study of the formation of aerenchyma (cortical gas-filled space) in adventitious roots of rice (Oryza sativa), J Exp Bot 37: 832–841
Wittenbach VA, Lin W, Hebert RR (1982) Vacuolar localization of proteases and degradation of chloroplasts in mesophyll protoplasts from senescing primary wheat leaves. Plant Physiol 69: 98–102
Yamasaki T, Kudoh T, Kamimura Y, Katoh S (1996) A vertical gradient of the chloroplast abundance among leaves ofChenopodium album. Plant Cell Physiol 37: 43–48
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Inada, N., Sakai, A., Kuroiwa, H. et al. Senescence program in rice (Oryza sautiva L.) leaves: Analysis of the blade of the second leaf at the tissue and cellular levels. Protoplasma 207, 222–232 (1999). https://doi.org/10.1007/BF01283003
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01283003