Summary
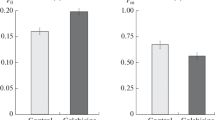
The colchicine-binding activity in carrot-cell extract was markedly enhanced and also stabilized by the addition of tartrate. The optimum temperature for the binding reaction in the presence of tartrate shifted to 41 °C from 30 °C in its absence. The apparent affinity constant in the presence of tartrate was 1.6 × 105l/mol at 37 °C which is much higher than the value obtained in the absence of tartrate. It was confirmed from the following evidence that the binding reaction in the presence of tartrate represented the colchicinetubulin interaction. 1. The reaction was inhibited by the preincubation with proteases and not with lumicolchicine. 2. The colchicine-binding component had a similar property to porcine brain tubulin in DEAE-Sephacel column chromatography and SDSpolyacrylamide gel electrophoresis.
Similar content being viewed by others
References
Arakawa, T., Timasheff, S. N., 1982 a: Preferential interactions of proteins with salts in concentrated solutions. Biochemistry21, 6545–6552.
— —, 1982 b: On protein-structure stabilizing solvent. Seikagaku54, 1307–1310.
Bhattacharyya, B., Wolff, J., 1975: Membrane-bound tubulin in brain and thyroid tissue. J. biol. Chem.250, 7639–7646.
— —, 1976: Anion-induced increases in the rate of colchicine binding to tubulin. Biochemistry15, 2283–2288.
Hamel, E., Lin, C. M., 1981: Stabilization of the colchicine-binding activity of tubulin by organic acids. Biochim. biophys. Acta675, 226–231.
Hart, J. W., Sabnis, D. D., 1976: Colchicine binding activity in extracts of higher plants. J. exp. Bot.27, 1353–1360.
Heath, I. B., 1975: Colchicine and colcemid binding components of the fungusSaprolegnia ferax. Protoplasma85, 177–192.
Ledbetter, M. C., Porter, K. R., 1970: Introduction to the fine structure of plant cells. Berlin-Heidelberg-New York: Springer.
Lee, J. C., Timasheff, S. N., 1981: The stabilization of proteins by sucrose. J. biol. Chem.256, 7193–7201.
Lowry, P. E., Rosebrough, N. J., Farr, A. L., Randall, R. J., 1951: Protein measurement with the Folin phenol reagent. J. biol. Chem.193, 265–275.
Mizuno, K., Koyama, M., Shibaoka, H., 1981: Isolation of plant tubulin from azuki bean epicotyl by ethyl N-phenyl-carbamateSepharose affinity chromatography. J. Biochem.89, 329–332.
Morejohn, L. C., Fosket, D. E., 1982: Higher plant tubulin identified by self-assembly into microtubulesin vitro. Nature297, 426–428.
Murashige, T., Skooo, F., 1962: A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant.15, 473–497.
Ogita, Z., Markert, C. L., 1979: A miniaturized system for electrophoresis on polyacrylamide gels. Anal. Biochem.99, 233–241.
Okamura, S., 1980: Binding of colchicine to a soluble fraction of carrot cells grown in suspension culture. Planta149, 350–354.
Rikin, A., Atsmon, D., Gitler, C., 1982: Extraction and immunochemical assays of a tubulin-like factor in cotton seedlings. Planta154, 402–406.
Scatchard, G., 1949: The attractions of proteins for small molecules and ions. Ann. N. Y. Acad. Sci.51, 660–672.
Shelanski, M. L., Gaskin, F., Canter, C. R., 1973: Microtubule assembly in the absence of added nucleotides. Proc. Natl. Acad. Sci. U.S.A.70, 765–768.
Solomon, F., Monard, D., Rentsch, M., 1973: Stabilization of colchicine-binding activity of neuroblastoma. J. mol. Biol.78, 569–573.
Weber, K., Osborn, M., 1969: The reliability of molecular weight determinations by dodecyl sulfate-acrylamide gel electrophoresis. J. biol. Chem.244, 4406–4412.
Weisenberg, R. C., Borisy, G. G., Taylor, E. W., 1968: The colchicine-binding protein of mammalian brain and its relation to microtubules. Biochemistry12, 4466–4479.
Wilson, L., 1970: Properties of colchicine-binding protein from chick embryo brain. Interactions with vinca alkaloids and podophyllotoxin. Biochemistry9, 4999–5007.
—,Bryan, J., 1974: Biochemical and pharmacological properties of microtubules. Adv. Cell molec. Biol.3, 21–72.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Okamura, S. Effect of tartrate on the colchicine-binding activity in cultured carrot cell extract. Protoplasma 118, 199–205 (1983). https://doi.org/10.1007/BF01281803
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01281803