Summary
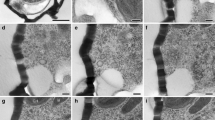
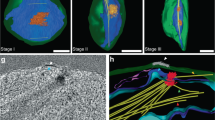
Microtubule systems appear sequentially at the distal and proximal poles of tetrad members during mid-sporogenesis in the mossTetraphis pellucida Hedw. The distal microtubule system emanates from a microtubule organizing center (MTOC) located between the single plastid and the nucleus. The distal MTOC and associated microtubules, which appear immediately after cytokinesis, are ephemeral and do not appear to be associated with the deposition of exine occuring at the same time. The proximal microtubule system, which appears slightly later than the distal system, is a more stable component of mid-sporogenesis. The proximal MTOC is an irregularly lobed, patelliform aggregation of electron-dense granules located beneath the plasma membrane at the proximal spore pole. Several bundles of microtubules radiate from the proximal MTOC and traverse the cell, enclosing the nucleus in an cone of microtubules. The proximal microtubule system is thought to function in aperture development and organelle migration. The relatively large nucleus migrates a short distance in the small spore early in the tetrad stage and maintains its acentric position at the proximal pole throughout later stages of sporogenesis. The plastid migrates later in the tetrad stage from its meiotic position parallel to the distal surface to a position perpendicular to the distal surface with one tip in close proximity to the proximal MTOC. The proximal microtubule system reaches its maximum development by the end of the tetrad stage and all micrographic evidence of it is lost in the maturation stages of late sporogenesis.
Similar content being viewed by others
References
Bassel, A. R., Kuehnert, C. C., Miller, J. H., 1981: Nuclear migration and asymmetric cell division inOnoclea sensibilis spores: an ultrastructural and cytochemical study. Amer. J. Bot.68, 350–360.
Brown, R. C., Lemmon, B. E., 1980 a: Ultrastructure of sporogenesis in a mossDitrichum pallidum. I. Meiotic prophase. Bryologist83, 137–152.
—, 1980 b: Ultrastructure of sporogenesis in a moss,Ditrichum pallidum. II. Metaphase I through the tetrad. Bryologist83, 153–160.
—, 1980 c: Ultrastructure of sporogenesis in a moss,Ditrichum pallidum. III. Spore wall formation. Amer. J. Bot.67, 918–934.
—, 1981: Aperture development in spores of the mossTrematodon longicollis Mx. Protoplasma106, 273–287.
—, 1982 a: Ultrastructure of meiosis in the mossRhynchostegium serrulatum. I. Prophasic microtubules and spindle dynamics. Protoplasma110, 23–33.
—, 1982 b: Ultrastructural aspects of moss meiosis: Review of nuclear and cytoplasmic events during prophase. J. Hattori Bot. Lab.53, 29–39.
Fowke, L. C., Pickett-Heaps, J. D., 1978: Electron microscope study of vegetative cell division in two species ofMarchantia. Canad. J. Bot.56, 467–475.
Galatis, B., 1980: Microtubules and guard cell morphogenesis inZea mays L. J. Cell Sci.45, 211–244.
Gantt, E., Arnott, H. J., 1965: Spore germination and development of the young gametophyte of the ostrich fern (Matteuccia struthiopteris). Amer. J. Bot.52, 82–94.
Gunning, B. E. S., 1981: Microtubules and cytomorphogenesis in a developing organ: the root primordium ofAzolla pinnata. In: Cytomorphogenesis in plants (Kiermayer, O., ed.), pp. 301–325. Wien and New York: Springer.
Hepler, P. K., 1981: Morphogenesis of tracheary elements and guard cells. In: Cytomorphogenesis in plants (Kiermayer, O., ed.), pp. 327–347. Wien and New York: Springer.
Huckaby, C. S., Raghavan, V., 1981 a: Spore germination patterns in the ferns,Cyathea andDicksonia. Ann. Bot.47, 297–401.
—, 1981 b: The spore-germination pattern of Thelypteroid ferns. Amer. J. Bot.68, 517–523.
Kiermayer, O., 1972: Beeinflussung der postmitotischen Kernmigration vonMicrasterias denticulata Breb. durch das Herbizid Trifluralin. Protoplasma75, 421 -426.
Kreitner, G. L., Carothers, Z. B., 1976: Studies of spermatogenesis in the Hepaticae V. Blepharoplast development inMarchantia polymorpha. Amer. J. Bot.63, 545–557.
Lambert, A.-M., 1974: Ultrastructure de l'appareil fusorial en méiose chez la mousseMnium hornum L. Bull. Soc. Bot. France121, 93–96.
—, 1978: Laméiose chezles Bryophytes: ultrastructure et dynamique du fuseau chez une mousse,Mnium hornum Hedw. Bryophyt. Biblio.13, 113–145.
Marchant, H. J., 1979: Microtubular determination of cell shape during colony formation by the algaPediastrum. Protoplasma98, 1–14.
Mueller, D. M. J., 1974: Spore wall formation and chloroplast development during sporogenesis in the mossFissidens limbatus. Amer. J. Bot.61, 525–534.
Palevitz, B. A., 1981: Microtubules and possible microtubule nucleation centers in the cortex of stomatal cells as visualized by high voltage electron microscopy. Protoplasma107, 115–125.
Pickett-Heaps, J. D., Fowke, L. C., 1970: Mitosis, cytokinesis, and cell elongation in the desmid,Closterium littorale. J. Phycol.6, 189–215.
Raghavan, V., Huckaby, C. S., 1980: A comparative study of cell division patterns during germination of spores ofAnemia, Lygodium andMohria (Schizaeaceae). Amer. J. Bot.67, 653–663.
Schmiedel, G., Schnepf, E., 1979 a: Side branch formation and orientation in the caulonema of the moss,Funaria hygrometrica: normal development and fine structure. Protoplasma100, 367–389.
—, 1979 b: Side branch formation and orientation in the caulonema of the moss,Funaria hygrometrica: experiments with inhibitors and with centrifugation. Protoplasma101, 47–59.
Stearns, M. E., Brown, D. L., 1981: Microtubule organizing centers (MTOCs) of the algaPolytomella exert spatial control over microtubule initiationin vivo andin vitro. J. Ultrastruct. Res.77, 366–378.
Vogelmann, Th. C., Bassel, A. R., Miller, J. H., 1981: Effects of microtubule-inhibitors on nuclear migration and rhizoid differentiation in germinating fern spores. Protoplasma109, 295–316.
Woodcock, C. L. F., 1971: The anchoring of nuclei by cytoplasmic microtubules inAcetabularia. J. Cell Sci.8, 611–621.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brown, R.C., Lemmon, B.E. Microtubule organization and morphogenesis in young spores of the mossTetraphis pellucida Hedw. Protoplasma 116, 115–124 (1983). https://doi.org/10.1007/BF01279828
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01279828