Summary
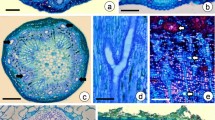
The development and structure of sclerotia of the fungusSclerotinia minor Jagger, was studied by light, scanning and transmission electron microscopy. The sclerotia formed beneath a weft of overlying vegetative hyphae, that sometimes became enveloped as the sclerotia enlarged. Differentiation of the sclerotial hyphae into regions of rind, cortex and medulla, began only 12 to 24 hours after sclerotial initiation occurred. The cortex was the last region to become discernible. The rind cells rapidly became vacuolate, while their walls thickened and became pigmented. At maturity the rind consisted of a closely packed layer of cells around the sclerotium. The cortex was about three cells wide and was made up of pseudoparenchymatous tissue. The prosenchymatous medulla constituted the main part of the sclerotium. Cytoplasmic reserves, tentatively identified as polyphosphate granules and protein bodies, accumulated in large numbers in cortical and medullary hyphae. Extracellular material was laid down very rapidly around hyphae of the cortex and medulla, until at maturity, it almost completely filled any interhyphal spaces. The ultrastructure of young sclerotial hyphae was very similar to that of actively growing vegetative hyphae. The numbers of nuclei and profiles of mitochondria decreased at later stages of development but there was an increase in the number of profiles of endoplasmic reticulum cisternae. The cytoplasm had a granular appearance throughout differentiation. The general structure of mature sclerotia ofS. minor was similar to that reported for sclerotia of other species in the genusSclerotinia.
Similar content being viewed by others
References
Briarty, L. G., Coult, D. A., Boulter, D., 1969: Protein bodies of developing seeds ofVicia faba. J. exp. Bot.20, 358–372.
Bussel, J, Sommer, N. F., 1973: Lomasome development inRhizopus stolonifer sporangiophores during anaerobiosis. Can. J. Microbiol.19, 905–907.
Bullock, S., Ashford, A. E., Willetts, H. J., 1980: The structure and histochemistry of sclerotia ofSclerotinia minor Jagger II. Histochemistry of extracellular substances and cytoplasmic reserves. Protoplasma104, 333–351.
Colotelo, N., 1974: A scanning electron microscope study of developing sclerotia ofSclerotinia sclerotiorum. Can. J. Bot.52, 1127–1130.
Cooke, R. C., 1971: Physiology of sclerotia ofSclerotinia sclerotiorum during growth and maturation. Trans. Br. mycol. Soc.56, 51–59.
Corner, E. J. H., 1950: A monograph ofClavaria and allied genera. London: Oxford University Press.
Feder, N., O'Brien, T. P., 1968: Plant microtechnique: some principles and new methods. Am. J. Bot.55, 123–142.
Fineran, B. A., Bullock, S., 1972: A procedure for embedding plant material in Araldite for electron microscopy. Ann. Bot.36, 83–86.
—,Lee, M. S. L., 1975: Organization of quadrifid and bifid hairs in the trap ofUtricularia monanthos. Protoplasma84, 43–70.
Harold, F. M., 1966: Inorganic polyphosphates in biology: structure, metabolism, and function. Bact. Rev.30, 772–794.
Ingold, C. T., 1959: Jelly as a water-reserve in fungi. Trans. Br. mycol. Soc.42, 475–478.
Jensen, T. E., 1968: Electron microscopy of polyphosphate bodies in a blue-green alga,Nostoc pruniforme. Arch. Mikrobiol.62, 144–152.
Khan, S. R., Aldrich, H. C., 1973: Conidiogenesis inTermitaria snyderi (Fungi Imperfecti). Can. J. Bot.51, 2307–2314.
Kohn, L. M., 1979: A monographic revision of the genusSclerotinia. Mycotaxon9, 365–444.
Marchant, R., Peat, A., Bunbury, G. H., 1967: The ultrastructural basis of hyphal growth. New Phytol.66, 623–629.
—,Robards, A. W., 1968: Membrane systems associated with the plasmalemma of plant cells. Ann. Bot.32, 457–471.
O'Brien, T. P., 1972: The cytology of cell-wall formation in some eukaryotic cells. Bot. Rev.38, 87–118.
Purdy, L. H., 1955: A broader concept of the speciesSclerotinia sclerotiorum based on variability. Phytopathology45, 421–427.
Rest, J. A., Vaughan, J. G., 1972: The development of protein and oil bodies in the seed ofSinapis alba L. Planta105, 245–262.
Saito, I., 1974: Ultrastructural aspects of the maturation of sclerotia ofSclerotinia sclerotiorum (Lib.) de Bary. Trans. mycol. Soc. Japan15, 384–400.
—, 1977: Studies on the maturation and germination of sclerotia ofSclerotinia sclerotiorum (Lib.) de Bary, a causal fungus of bean stem rot. Rep. Hokkaido Prefect. Agric. Expt. Sta.26, 1–106.
White, J. A., Brown, M. F., 1979: Ultrastructure and X-ray analysis of phosphorous granules in a vesicular-arbuscular mycorrhizal fungus. Can. J. Bot.57, 2812–2818.
Willetts, H. J., Wong, A. L., 1971: Ontogenetic diversity of sclerotia ofSclerotinia sclerotiorum and related species. Trans. Br. mycol. Soc.57, 515–524.
—,Wong, J. A.-L., 1980: The biology ofSclerotinia sclerotiorum, S. trifoliorum, andS. minor with emphasis on specific nomenclature. Bot. Rev.46, 101–165.
Wolf, F. A., 1958: Mechanism of apothecial opening and ascospore expulsion by the cupfungusUrnula craterium. Mycologia50, 837–843.
Wong, A.-L., Willetts, H. J., 1975: A taxonomic study ofSclerotinia sclerotiorum and related species: mycelial interactions. J. gen. Microbiol.88, 339–344.
Author information
Authors and Affiliations
Additional information
Mrs. S.Lowry.
Rights and permissions
About this article
Cite this article
Bullock, S., Willetts, H.J. & Ashford, A.E. The structure and histochemistry of sclerotia ofSclerotinia minor Jagger. Protoplasma 104, 315–331 (1980). https://doi.org/10.1007/BF01279776
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01279776