Summary
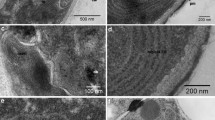
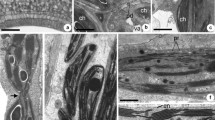
An ultrastructural study of the fungal plant pathogen,Venturia inaequalis (Cke)Wint., has revealed the presence of junctions that form within germinated ascospores as they penetrate the cuticle of apple leaves. This is the first report of junctional structures in fungi or in any organism with a cell wall. Morphologically they resemble septate junctions of invertebrate tissues, but developmentally they differ in origin. Junctions inV. inaequalis form intracellularly during formation of the infection sac by an invagination and subsequent folding back of the plasma membrane around the penetration pore. The apposed plasma membranes form junctions and result in a junctional belt around the penetration pore. The plasma membrane and the junctional structures stain positively with phosphotungstic acid and the confluent infection sac membrane remains unstained. As the infection sac grows, additional junctions form between the infection sac membrane and plasma membrane. The intermembrane space of these junctions measures 17 nm, and the septa occur at regular 2.5 nm intervals. The junctions appear to provide adhesive support and structural rigidity for the developing infection sac and may also facilitate differentiation between the infection sac membrane and the plasma membrane. Similarities and differences between these junctions and invertebrate septate junctions are discussed.
Similar content being viewed by others
Abbreviations
- A :
-
ascospore
- cu :
-
cuticle
- fw :
-
fungal wall
- IS :
-
infectionsac
- mu :
-
mucilage
- P :
-
Penetration pore
- pl :
-
plasma membrane
- pw :
-
plant wall
- w :
-
epicuticular wax
References
Dragsten PR, Blumenthal R, Handler JS (1981) Membrane asymmetry in epithelia: is the tight junction a barrier to diffusion in the plasma membrane? Nature 294: 718–722
Green CR, Bergquist PR (1982) Phylogenetic relationships within the invertebrata in relation to the structure of septate junctions and the development of “occluding” junctional types. J Cell Sci 53: 279–305
Kausch AP, Horner HT (1984) Differentiation of raphide crystal idioblasts in isolated root culture ofYucca torreyi (Agavaceae). Can J Bot 62: 1474–1484
Lane NJ, Skaer H Le B (1980) Intercellular junctions in the central nervous system of insects. J Cell Sci 26: 175–199
Maeda KM (1970) An ultrastructural study ofVenturia inaequalis (Cke) Wint. Infection ofMalus host. M. Sc. Thesis, Purdue University, Lafayette, Indiana
Mollenhauer HH (1964) Plastic embedding mixtures for use in electron microscopy J Stain Technol 39: 111–114
Nicholson RL, Kuc J, Williams EB (1972) Histochemical demonstration of transitory esterase activity inVenturia inaequalis. Phytopathology 62: 1242–1247
Noirot-Timothee C, Noirot C (1980) Septate and scalaroform junctions of the arthropods. Int Rev Cytol 63: 97–140
Reinhardt C, Hecker H (1973) Structure and function of the basal lamina and of the cell junctions in the midgut epithelium (stomach) of femaleAedes aegypti L. (Insecta, Diptera). Acta Trop 30: 213–235
Roland JC, Lembi CA, Morre DJ (1972) Phosphotungstic acid-chromic acid as a selective electron-dense stain for plasma membranes of plant cells. Stain Technol 47: 195–200
Smereka KJ,MacHardy WE,Kausch AP (1987) Cellular differentiation ofVentura inaequalis ascospores during germination and penetration of apple leaves. Accepted with revision. Can J Bot
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26: 31–34
Staehelin LA (1974) Structure and function of intercellular junctions. Int Rev Cytol 39: 191–283
—,Hull BE (1978) Junctions between living cells. Scient. Am. 238: 140–152. Membranes of plant cells. Stain Technol 47: 195–200
Thiéry JP (1967) Mise en evidence des polysaccharies sur coupes fines en microscopie electronique. J Microsc (Paris) 6, 987–1018
Turner ML, MacHardy WE, Gadoury DM (1986) Germination and appressorium formation byVenturia inaequalis during infection of apple seedling leaves. Plant Disease 70: 658, 660
Van Meer G, Gumbiner B, Simon K (1986) The tight junction does not allow lipid molecules to diffuse from one epithelial cell to the next. Nature 322, 639–641
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smereka, K.J., Kausch, A.P. & MacHardy, W.E. Intracellular junctional structures in germinating ascopores ofVenturia inaequalis . Protoplasma 142, 1–4 (1988). https://doi.org/10.1007/BF01273220
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01273220