Summary
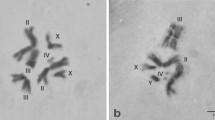
The salivary gland nuclei of larvalGlyptotendipes barbipes from Stratford (Ontario) comprise 3 pairs of long metacentric chromosomes (I–III) and one pair of short acrocentrics (IV). Homologues are closely paired when homozygous, centromeres are expressed as heterochromatic drums. Each chromosome carries a nucleolus, and Chromosome IV in addition carries a Ring of Balbiani. The gross polytene idiogram is identical with that implicit inBauer's cytological analysis of the same species from Germany.
In direct comparison the banding pattern of the German and Canadian larvae proved identical for at least one sequence in each chromosome arm.
Three simple inversions are known in the species; one (III L-1) is endemic to Germany, a second (II L-3) has been found only once in Canada and a third (I S-1) is common in Canada but has not been reported from Germany. The remaining 2 inversions (II L-1,2 and III S-1,2) are complex and are here analysed as included types. Rearrangement II L-1,2 occurs both in Canada and in Germany and achieves heterozygous frequencies near 50% in both countries. It is the first inversion reported to be holarctic in distribution in insects not associated with man. The second complex inversion III S-1,2 has been found only in Canada where it is common. In each of the complex inversions at least one pair of breaks is near coincident.
Meiosis is normal and chiasmatic in structurally homozygous males. It lacks conspicuous localization of chiasmata. No meiotic abnormalities were observed in inversion heterozygotes. It is postulated that complex heterozygous inversions interfere with pairing to such an extent that the residual ill-effects are out balanced by heterosis of co-adapted systems.
A selective advantage of heterozygosis is directly indicated for II L by a significant (p=0.02) excess of II L/II L-1,2 larvae over the Hardy-Weinberg expectation.
Similar content being viewed by others
References
Bauer, H.: Beiträge zur vergleichenden Morphologie der Speicheldrüsenchromosomen (Untersuchungen an den Riesenchromosomen der Dipteren II). Zool. Jb., Abt. allg. Zool.56, 239–276 (1936).
—: Fortschr. Zool. Cytologie2, 12 (1937).
Beermann, W.: Cytologische Analyse einesCamptochironomus-Artbastards. Chromosoma7, 198–259 (1955).
Breuer, M.E., u.C. Pavan: Behaviour of polytene chromosomes ofRhynchosciara angelae at different stages of larval development. Chromosoma7, 371–386 (1955).
Chu, J.: Reaction of nucleic acid to acetocarmine. Nature157, 513 (1946).
Conger, A.D., andL.M. Fairchild: A quickfreeze method for making smear slides permanent. Stain Technol.28, 281–283 (1953).
Hsu, T. C., andT.T. Liu: Microgeographic analysis of chromosomal variation in a Chinese species ofChironomus (Diptera). Evolution2, 49–57 (1948).
Mainx, F.: Der chromosomale Strukturpolymorphismus natürlicher Populationen als Problem der Genetik und der Evolution. Instituto Gregorio Mendel, Roma, Mendel-Festschrift. 1955.
Mainx, F., E. Kunze u.Th. Koske: Cytologische Untersuchungen an Lunzer Chironomiden. Österr. Zool. Z.4, 33–44 (1953).
Novitski, E.: Chromosome variation inDrosophila athabasca. Genetics31, 508–524 (1946).
Rothfels, K.H., andW. Fairlie: The nonandom distribution of inversion breaks in the midgeTendipes decorus. Canad. J. Zool.35, 221–263 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Basrur, V.R. Inversion polymorphism in the midge Glyptotendipes barbipes (Staeger). Chromosoma 8, 597–608 (1956). https://doi.org/10.1007/BF01259521
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01259521