Summary
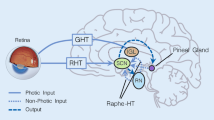
To determine whether extra-retinal mechanisms mediate photoperiodic changes in neonatal rat pineal gland N-acetyltransferase activity, 4-day-old intact or bilateral orbital enucleated rats were killed during the dark phase of the lighting cycle, either in darkness or following 4 hr exposure to fluorescent light. Light suppressed the high nighttime N-acetyltransferase activity equally in intact and enucleated pups. Subsequent studies showed that at least 0.5 hr exposure and nocturnal illuminances of 109 ΜW/cm2 or greater were required to cause statistically significant reductions in the activity of the enzyme in 4-day-old rats. Taken together, these data indicate that relatively intense environmental light can affect neonatal rat pineal gland biochemistry via extra-retinal mechanisms.
Similar content being viewed by others
References
Altar A, Motroni T, Lytle LD (1983) Functional synaptogenesis and the rat pineal gland: a pharmacological investigation. J Neural Transm 58: 231–244
Binkley S, MacBride SE, Klein DC, Ralph CK (1973) Pineal enzymes: regulation of avian melatonin synthesis. Science 181: 273–275
Bridges CDB, Foster RG, Landers RA, Fong SL (1987) Interstitial retinol-binding protein and cellular retinol-binding protein in the mammalian pineal. Vision Res 27: 2049–2060
Bronstein DM, Haak KA, Torres G, Lytle LD (1985) Light pulse inhibition of pineal gland N-acetyltransferase: developmental aspects. Fed Proc 44: 998
Bucana CD, Nadakavukaren NJ (1973) Ultrastructural investigation of the postnatal development of the hamster Harderian gland. II. Male and female. Z Zellforsch 142: 1–12
Deguchi T, Axelrod J (1972) Sensitive assay for serotonin N-acetyltransferase in rat pineal. Anal Biochem 50: 174–179
Deguchi T (1979) Circadian rhythm of serotonin N-acetyltransferase activity in organ culture of chick pineal gland. Science 203: 1245–1247
Ellison N, Weller JL, Klein DC (1972) Development of a circadian rhythm in the activity of pineal serotonin N-acetyltransferase. J Neurochem 19: 1335–1341
Ganong WF, Shepherd MD, Wall JR, Brunt EE, Van Clegg MT (1963) Penetration of light into the brain of mammals. Endocrinology 72: 962–963
Hill RM (1947) The control of body temperature in white rats. Am J Physiol 149: 650–656
Hoffman RA, Corth R (1988) Photic environments: physical and biological considerations. Pineal Res Rev 6: 95–118
Kalsow CM, Wacker WB (1977) Pineal reactivity of anti-retina sera. Invest Ophthalmol Vis Sci 16: 181–184
Klein DC, Moore RY (1979) Pineal N-acetyltransferase and hydroxyindole-O-methyltransferase: control by the retinohypothalamic tract and the suprachiasmatic nucleus. Brain Res 174: 245–262
Klein DC, Auerbach DA, Namboodiri MAA, Wheeler GHT (1981) Indole metabolism in the mammalian pineal gland. In: Reiter RJ (ed) The pineal gland: anatomy and biochemistry. CRC, Boca Raton, Fl, pp 199–227
Korf HW, Oksche A (1986) Photoneuroendocrine aspects of the pineal gland: phylogeny and ontogeny. In: Gupta D, Reiter RJ (eds) The pineal gland during development: from fetus to adult. Croom Helm, London, pp 1–13
Lytle LD, Keil FC (1974) Brain and peripheral monoamines: possible role in the ontogenesis of normal and drug-induced responses in the immature mammal. In: Fuxe K, Olson L, Zotterman Y (eds) Dynamics of degeneration and growth in neurons. Pergamon Press, New York, pp 575–591
Lytle LD, Haak KA, Torres G (1988) Maturational changes in the rat pineal gland and the neural mechanisms controlling its functions. Pineal Res Rev 6: 118–172
Machado CRS, Machado ABM, Wragg LE (1969) Circadian serotonin rhythm control: sympathetic and nonsympathetic pathways in rat pineals of different ages. Endocrinology 85: 846–849
Moore RY, Klein DC (1974) Visual pathways and the central neural control of a circadian rhythm in pineal serotonin N-acetyltransferase activity. Brain Res 71: 17–33
Oksche A (1984) Evolution of the pineal complex: correlation of structure and function. Ophthalmic Res 16: 88–95
Sakai T (1981) The mammalian Harderian gland: morphology, biochemistry, function, and phylogeny. Arch Histol Jpn 44: 299–333
Sommers RL, Klein DC (1984) Rhodopsin kinase activity in the mammalian pineal gland and other tissues. Science 226: 182–184
Torres G, Haak KA, Lytle LD (1989) Catecholaminergic mechanisms mediate hypothermiainduced elevations in pineal gland N-acetyltransferase in neonatal rats. J Pineal Res 6: 43–53
Vanecek J, Illnerova H (1985) Effect of short and long photoperiods on pineal N-acetyltransferase rhythm and on growth of testes and brown adipose tissue in developing rats. Neuroendocrinology 41: 186–191
Wetterberg L, Geller E, Yuwiler A (1970) Harderian gland: an extraretinal photoreceptor influencing the pineal gland in neonatal rats? Science 167: 884–885
Yuwiler A, Klein DC, Buda M, Weller JL (1977) Adrenergic control of pineal N-acetyltransferase activity: developmental aspects. Am J Physiol 233: E141-E146
Zimmerman BL, Tso MO (1975) Morphological evidence of photoreceptor differentiation of pinealocytes in the neonatal rat. J Cell Biol 66: 60–75
Zweig M, Snyder SH, Axelrod J (1966) Evidence for a nonretinal pathway of light to the pineal gland of newborn rats. Proc Natl Acad Sci USA 56: 515–520
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Torres, G., Lytle, L.D. Light affects neonatal rat pineal gland N-acetyltransferase activity by an extra-retinal mechanism. J. Neural Transmission 80, 67–77 (1990). https://doi.org/10.1007/BF01245023
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01245023