Abstract
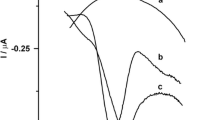
A bentonite-modified carbon paste electrode has been applied to the determination of 2-nitrophenol by differential pulse voltammmetry. The electrochemical reduction of 2-nitrophenol at −0.8 V is carried out in an artificial sea water-formic acid/sodium formate medium at pH 4. The peak height was found to be dependent on the pH over the range 2–11; the presence of a secondary process was observed in the pH range 8–11. The peak potential showed a dependence on pH, with two linear regions with different slopes. A linear relationship between peak intensity and concentration was obtained in the range 0.07–10 mgl−1, with a detection limit of 0.03 mg 1−1 and a coefficient of variation of 1.3% at 5 mg 1−1. The effects of organic and inorganic species on the 2-nitrophenol determination were studied with a view to testing the resolution of the voltammetric technique. The proposed method has been applied to sea water samples with good results.
Similar content being viewed by others
References
U. S. Environmental Protection Agency,Fed. Regist. 1979,44, 233.
U. S. Environmental Protection Agency,Fed. Regist. 1989,52, 131.
P. A. Realini,J. Chromatogr. Sci. 1981,19, 124.
U. S. Environmental Protection Agency,Quality Assurance and Quality Control for Screening and Verification of Industrial Effluents for Prioritary Pollutants, Environmental Monitoring and Support Laboratories, Cincinnati, 1979, 45268.
U. S. Environmental Protection Agency,Fed. Regist. 1973,38, 125.
D. N. Armentrout, in:Chromatographic Analysis of the Environment, 2nd Ed. (R. L. Grob ed.), Marcel Dekker, New York, 1983, pp. 569–570.
L. Michel, A. Zatka,Anal. Chim. Acta 1979,105, 109.
A. Trojanek, H. G. De Jong,Anal. Chem. 1982,141, 115.
J. J. Scanlon, P. A. Flaquer, G. W. Robinson, G. E. O'Brien, P. E. Sturrock,Anal. Chim. Acta 1984,158, 169.
M. Zietek,Mikrochim. Acta 1975,II, 463.
H. Burgschat, K. J. Netter,J. Pharm. Sci. 1977,66, 60.
T. Matsue, M. Fujihira, T. Osa,Anal. Chem. 1981,53, 722.
J. Barek, H. Ebertová, V. Mejstrik, J. Zima,Collect. Czech. Chem. Commun. 1994,59, 1761.
L. Hernández, P. Hernández, J. Vicente,Fresenius J. Anal. Chem. 1993,345, 712.
E. Álvarez, M. T. Sevilla, J. M. Pinilla, L. Hernández,Anal. Chim. Acta 1992,260, 19.
R. Agraz, M. T. Sevilla, J. M. Pinilla, L. Hernández,Electroanalysis 1991,393, 3.
P. Hernández, E. Alda, L. Hernández,Fresenius Z. Anal. Chem. 1987,327, 676.
L. Hernández, P. Hernández, M. H. Blanco, E. Lorenzo,Analyst 1988,113, 1719.
L. Hernández, P. Hernández, E. Lorenzo, in:Contemporary Electroanalytical Chemistry (A. Ivaska, A. Lewenstam, R. Sara eds.), Plenum, New York, 1990, p. 205.
K. Stulik, V. Pacakova,Electroanalytical Measurements in Flowing liquids, Ellis Horwood, Chichester, 1987, pp. 236 and 269.
L. F. Fieser, M. Fieser,Qumicia Orgánica Superior, Ediciones Grijalbo, Barcelona, 1966, p. 1296.
N. L. Allinger, M. P. Cava, D. C. De Jongh, C. R. Johnson, N. A. Lebel, C. L. Stevens,Quimica Orgánica, 2nd Ed., Editorial Reverté, Barcelona, 1981, p. 810.
L. H. Keith, W. Crummett, J. Deegan, R. A. Libby, J. K. Taylor, G. Wentler,Anal. Chem. 1983,55, 2210.
J. C. Miller, J. N. Miller,Estadistica para Quimica Analitica, 2nd Ed., Addison-Wesley Iberoamericana, Wilmington, 1993, pp. 100–102.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rodriguez, I.N., Zamora, M.B., Salvador, J.M.B. et al. Voltammetric determination of 2-nitrophenol at a bentonite-modified carbon paste electrode. Mikrochim Acta 126, 87–92 (1997). https://doi.org/10.1007/BF01242667
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01242667