Abstract
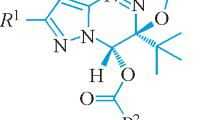
Oxidation of acetone-4-p-bromophenylsemicarbazone with lead tetraacetate yields only one isomer of 5,5-dimethyl-2-p-bromophenylimino-Δ3-1,3,4-oxadiazoline, C10H10BrN3O. Crystals of this compound are triclinic, space groupP¯1,a = 5·91(1),b = 9·52(1),c = 11·59(2) Å, α =114·1(1) °, β = 93·5(1) ° and γ = 108·8(1) °;z = 2 and the chemical composition is C10H10BrN3O. Of 2306 independent reflexions examined, 877 were strong enough to be measured with integrated precession photographs using MoKα radiation. The structure was refined using a full-matrix least-squares program to a finalR value of 0·064. The structure consists of planar phenyl and oxadiazoline rings, with a van der Waals interaction between a phenyl hydrogen atom and oxygen atom of the five member ring preventing the two rings from being co-planar; the H(13)-O(1) separation is 2·47 Å. TheZ-configuration of the product suggests a specific mechanism for the ring closure involving an organolead intermediate.
Similar content being viewed by others
References
Cameron, A. M., West, P. R. & Warkentin, J. (1969)J. Org. Chem. 34, 3230. Unpublished work of G. Gubelt, McMaster University.
International Tables for X-ray Crystallography (1962) Vol. III, Kynoch Press, Birmingham.
International Union of Pure and Applied Chemistry (1970)J. Org. Chem. 35, 2849 (rule E.2.2).
Layton, E. M., Jr., Kross, R. D. & Fassel, V. A. (1956)J. Chem. Phys. 25, 135.
Sadtler Standard Spectra, Sadtler Research Laboratories, Philadelphia, Pa.
West, P. R. & Warkentin, J. (1968)J. Org. Chem. 33, 2089.
Wheeler, A. S. (1929)J. Amer. Chem. Soc. 51, 3653.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Prakash, A., Calvo, C., Cameron, A.M. et al. Crystal structure of 5,5-dimethyl-2-p-bromophenylimino-Δ3-1,3,4-oxadiazoline. Journal of Crystal and Molecular Structure 3, 71–78 (1973). https://doi.org/10.1007/BF01237429
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01237429