Abstract
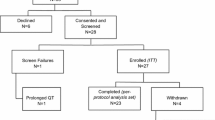
Formestane, a new selective aromatase inhibitor devoid of severe side-effects, has been shown to be active in patients with advanced breast cancer. To investigate the clinical activity and endocrinological effects of formestane as a first-line treatment, 52 patients were administered two different doses: 24 received 250 mg formestane and 28 received 500 mg formestane i.m. fortnightly. All of the patients had a performance status of 2 or less (ECOG scale), 34 (65%) had a disease-free interval of at least 2 years and 21 (40%) were both oestrogen-receptor- and progesterone-receptor-positive; 20 patients received hormone and 13 received chemotherapeutical adjuvant treatment. Objective responses were obtained in 8 patients in the 250-mg group (33%; 95% CI: 14%–52%) and in 13 patients in the 500-mg group (46%;, 95% CI.: 28%–64%). The median response duration in the two groups was respectively 11 and 12 months. E2 serum levels of oestradiol had significantly (P<0.001) decreased to more than 40% below the baseline value in both groups after 15 days of treatment, and remained unchanged thereafter. Local and systemic tolerability was satisfactory. We conclude that formestane is an effective and well-tolerated agent in previously untreated patients, and that these results should be confirmed by further studies.
Similar content being viewed by others
Abbreviations
- 17-OHCS :
-
17-hydroxycorticosteroids
References
Akhtar SS, Allan SG, Rodger A, Chetty UDI, Smyth JF, Leonard RCF (1991) A 10-year experience of tamoxifen as primary treatment of breast cancer in 100 elderly and frail patients. Eur J Surg Oncol 17: 30–35
Allan SG, Rodger A, Smyth JF, Leonard RCF, Chetty U, Forrest APM (1985) Tamoxifen as primary treatment of breast cancer in elderly or frail patients: a practical management. Br Med J 290: 358
Analysis of CRC Adjuvant Breast Trial (1988) Cyclophosphamide and tamoxifen as adjuvant therapies in the management of breast cancer. Br J Cancer 57: 604–607
Bajetta E, Zilembo N, Buzzoni R, Noberasco C, Celio L, Bichisao E (1993) Efficacy and tolerability of 4-hydroxyandrostenedione (4-OHA) as first-line treatment in postmenopausal patients with breast cancer after adjuvant therapy. Cancer Treat Rev 19: 31–36
Bajetta E, Zilembo N, Buzzoni R, Noberasco C, Di Leo A, Bartoli C, Merson M, Sacchini V, Moglia D, Celio L, Nelli P (1994) Endocrinological and clinical evaluation of two doses of formestane in advanced breast cancer. Br J Cancer 70: 145–150
Bradbeer JW, Kyngdon J (1983) Primary treatment of breast cancer in elderly women with tamoxifen. Clin Oncol 9: 31–34
Coombes RC, Huges SWM, Dowsett M (1992) 4-Hydroxyandrostenedione: a new treatment for postmenopausal patients with breast cancer. Eur J Cancer 28A: 1941–1945
Fisher B, Costantino J, Redmond C, Poisson R, Bowman D, Couture J et al from the National Surgical Adjuvant Breast and Bowel Project (NSABP) (1989) A randomized clinical trial evaluating tamoxifen in the treatment of patients with node-negative breast cancer who have estrogen-receptor-positive tumors. N Engl J Med 320: 479–484
Goss PE, Powles TJ, Dowsett M, Hutchison G, Brodie AMH, Etazet JC, Coombes RC (1986) Treatment of advanced postmenopausal breast cancer with an aromatase inhibitor, 4-hydroxyandrostenedione: phase II report. Cancer Res 46: 4823–4826
Hayward JL, Carbone PP, Heuson JC, Kumaoka S, Segaloff A, Rubens RD (1977) Assessment of response to therapy in advanced breast cancer. Eur J Cancer 13: 89–94
Höffken K, Jonat W, Possinger K, Kolbel M, Kunz T (1990) Aromatase inhibition with 4-hydroxyandrostenedione in the treatment of postmenopausal patients with advanced breast cancer: a Phase II study. J Clin Oncol 8: 875–880
Johnson PA, Bonomi PD, Anderson KM, Wolter JM, Economou SG (1986) Megestrol acetate: first line therapy for advanced breast cancer. Semin Oncol 13: 15–19
Johnston SRD, Dowsett M, Smith IE (1992) Towards a molecular basis for tamoxifen resistance in breast cancer. Ann Oncol 3: 503–511
Johnston SRD, Haynes BP, Smith IE, Jarman M, Sacks NPM, Ebbs SR, Dowsett M (1993) Acquired tamoxifen resistance in human breast cancer and reduced intra-tumoral drug concentration. Lancet 342: 1521–1522
Litherland S, Jackson IM (1988) Antiestrogens in the management of hormone-dependent cancer. Cancer Treat Rev 15: 183–194
Murphy D, West HF (1966) Urinary 17-hydroxycorticosteroids measured by gas-chromatography. J Endocrinol 36: 331–340
NATO Report (1988) Controlled trial of tamoxifen as a single adjuvant agent in the management of early breast cancer. Analysis at 8 years. Br J Cancer 57: 608–611
Peréz-Carríon R, Alberola Candel V, Calabresi F, Michel RTh, Santos R, Delozier T, Goss P, Mauriac L, Feuilhade F, Freue M, Pannuti F, Belle S van, Martinez J, Wehrle E, Royce CM (1994) Comparison of the selective aromatase inhibitor formestane with tamoxifen as first-line hormonal therapy in postmenopausal women with advanced breast cancer. Ann Oncol 5: S19-S24
Plowman PN (1993) Tamoxifen as adjuvant therapy in breast cancer. Current status. Drugs 46: 819–833
Powles TJ, Tillyer CR, Jones AL, Ashley SE, Treliaven J, Darvey JB, McKinna JA (1990) Prevention of breast cancer with tamoxifen — an update on the Royal Marsden Hospital Pilot Programme. Eur J Cancer 26: 680–684
Ravdin PM, Fritz NF, Tormey DC, Jordan VC (1988) Endocrine status of premenopausal node-positive breast cancer patients following adjuvant chemotherapy and long-term tamoxifen. Cancer Res 48: 1026–1029
Scottish Cancer Trials Office (1987) Report from the Breast Cancer Trials Committee. Adjuvant tamoxifen in the management of operable breast cancer: the Scottish trial. Lancet II: 172–175
Smalley RV, Sigal RB, Anderson S (1990) Front-line hormonal therapy for patients with advanced breast cancer: a community oncology research program. Semin Oncol 17: 47–51
Smith J, Harris AL, Morgan M, Ford T, Gazet JC, Harmer L, White H, Parsons A, Villardo A, Walsh G, McKinna J (1981) Tamoxifen versus aminoglutethimide in advanced breast carcinoma: a randomized cross-over trial. Br Med J 283: 1432–1434
Stein RC, Dowsett M, Hedley A, Devenport J, Gazet JC, Ford HT, Coombes RC (1990) Treatment of advanced breast cancer in postmenopausal women with 4-hydroxyandrostenedione. Cancer Chemother Pharmacol 26: 75–78
Trunet PF, Mueller PH, Girard F, Aupetit B, Bhatnagar AS, Zognbi F, Ezzet F, Menard J (1992) The effects of fadrozole hydrochloride on aldosterone secretion in healthy male subjects. J Clin Endocrinol Metab 74: 571–576
World Health Organization (1979) Handbook for reporting results of cancer treatment. WHO Offset publ 48
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zilembo, N., Bajetta, E., Noberasco, C. et al. Formestane: an effective first-line endocrine treatment for advanced breast cancer. J Cancer Res Clin Oncol 121, 378–382 (1995). https://doi.org/10.1007/BF01225692
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01225692