Summary
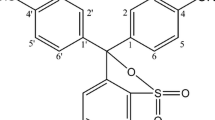
A procedure is described for the determination of 0.2–3.5 mg of the diphenyliodonium cation by extraction of its hexathiocyanatoferrate(III) Complex from slightly acidic solution into 1,2-dichloroethane. Absorbance measurements are made directly on the organic extract at 485 nm. Beer's law is not obeyed and a calibration curve must be used.
Zusammenfassung
Ein Verfahren für die Bestimmung von 0,2 bis 3,5 mg Diphenyljodoniumkation durch Extraktion seines Hexathiocyanatoferrat(III)-komplexes aus sehwach saurer Lösung mit 1,2-Dichloräthan wurde beschrieben. Die Absorption des organischen Extraktes wurde unmittelbar bei 485 nm gemessen. Das Beersche Gesetz ist nicht anwendbar, weshalb man eine Eichkurve verwenden muß.
Similar content being viewed by others
References
F. P. Dwyer andN. A. Gibson, Analyst76, 548 (1951).
M. Ziegler, O. Glemser, andN. Petri, Z. analyt. Chem.81, 154 (1957).
A. J. Bowd andD. Thorburn Burns, Mikrochim. Acta [Wien]1965, 1151.
F. M. Beringer, M. Drexler, E. M. Gindler, andC. C. Lumpkin, J. Amer. Chem. Soc.75, 2705 (1953).
P. Job, Ann. chim. (Paris)9, 113 (1928).
A. J. Bowd andD. Thorburn Burns, Mikrochim. Acta [Wien]1966, 1051.
Author information
Authors and Affiliations
Additional information
Part VI: Mikrochim. Acta [Wien]1967, 564. The work reported in this paper forms part of a thesis submitted byAnthony J. Bowd to the Faculty of Science of the University of London in partial fulfilment of the requirements for the degree of Doctor of Philosophy.
Rights and permissions
About this article
Cite this article
Bowd, A.J., Burns, D.T. Studies on the analytical chemistry of iodonium compounds. Part. VII. Mikrochim Acta 58, 1125–1129 (1970). https://doi.org/10.1007/BF01215947
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01215947