Abstract
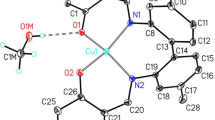
Bis(4-formyl-2-methoxyphenolato)bis(pyridine)copper(II) monohydrate crystallizes in the triclinic system:a = 10·432,b = 9·342,c = 13·799 Å, α = 103·75, β = 100·53, γ = 100·81 °,Z = 2, space groupP¯1.
The structure was determined by Fourier methods with Cukα diffractometer data, and refined by a combination of block-diagonal and full-matrix least squares toR = 0·068 for 2430 observed reflexions.
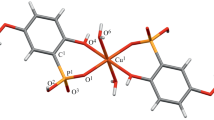
The copper atoms occupy the special positions 0, 0, 0 and 0,1/2, 1/2; hence, both molecules are centrosymmetric. Both the methoxy and phenolic oxygen atoms of the phenolate ions coordinate to the metal. These, together with coordinated nitrogen atoms from the pyridine molecules, give the copper atoms a six-fold coordination.
Hydrogen bonding between a water molecule and the phenolic oxygen atoms of adjacent molecules results in chains of molecules linked approximately along [011].
Similar content being viewed by others
References
Bullock, J. I., Hobson, R. J. & Povey, D. C. To be published.
Bullock, J. I. & Jones, S. L. (1970)J. Chem. Soc. (A) 2472.
Bullock, J. I. & Jones, S. L. (1971)J. Chem. Soc, (A) 2351.
Cromer, D. T. (1965)Acta Crystallogr. 18, 17.
Hanson, H. P., Herman, F., Lea, J. D. & Skillman, S. (1964)Acta Crystallogr. 17, 1040.
Hathaway, B. J. & Billing, D. E. (1970)Coord. Chem. Rev. (5), 143.
Pauling, L.Nature of the Chemical Bond, 3rd Ed. (Cornell Univ. Press, Ithaca, New York, 1960, p. 468).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hobson, R.J., Ladd, M.F.C. & Povey, D.C. Substituted phenols as ligands III: Crystal and molecular structure of bis(4-formyl-2-methoxyphenolato)bis(pyridine)copper(II) monohydrate. Journal of Crystal and Molecular Structure 3, 377–388 (1973). https://doi.org/10.1007/BF01203104
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01203104