Abstract
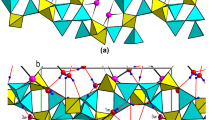
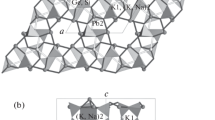
The crystal structure of octacalcium bis(hydrogenphosphate) tetrakis(phosphate)pentahydrate, Ca8(HPO4)2(PO4)4·5H2O, has been refined toR=0.051 for 4502 reflections. The crystals are triclinic:P¯1,a=19.692(4),b=9.523(2),c=6.835(2) Å,α=90.15(2),β=92.54(2), and γ=108.65(1)°,Z=2. The structure can be considered to consist of two layers parallel to (100), an apatitic layer which corresponds very closely to that of hydroxyapatite and a hydrated layer which contains all the water molecules. Several atoms in the hydrated layer are probably disordered. Possibilities for the hydrogen-bonding arrangement are discussed.
Similar content being viewed by others
References
Baur, W. H. (1974)Acta Crystallogr. B 30, 1195.
Berry, E. E., and Baddiel, C. B. (1967)Spectrochim Acta A 23, 1781.
Brown, W. E. (1962)Nature (London) 196, 1048.
Brown, W. E. (1966)Clin. Orthop. 44, 205.
Brown, W. E., Smith, J. P., Lehr, J. R., and Frazier, A. W. (1962)Nature (London) 196, 1050.
Brown, W. E., Mathew, M., and Tung, M. S. (1981)Prog. Cryst. Growth Charact. 4, 59.
Dickens, B., and Brown, W. E. (1969)Inorg. Chem. 8, 2093.
Donnay, G., and Allmann, R. (1970)Am. Miner. 55, 1003.
Finger, L. W., and Prince, E. (1975)Rfine4. Tech. Note 854. National Bureau of Standards, Washington, DC.
Fowler, B. O., Moreno, E. C., and Brown, W. E. (1966)Arch. Oral Biol. 11, 477.
International Tables for X-ray Crystallography (1974) Vol. IV (Kynoch Press, Birmingham), pp. 99–102.
Kay, M. I., Young, R. A., and Posner, A. S. (1964)Nature (London) 204, 1050.
Lenhert, P. G. (1975)J. Appl. Crystallogr. 8, 568.
Lippincott, E. R., and Schroeder, R. (1955)J. Chem. Phys. 23, 1099.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mathew, M., Brown, W.E., Schroeder, L.W. et al. Crystal structure of octacalcium bis(hydrogenphosphate) tetrakis(phosphate)pentahydrate, Ca8(HP04)2(PO4)4·5H2O. Journal of Crystallographic and Spectroscopic Research 18, 235–250 (1988). https://doi.org/10.1007/BF01194315
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01194315