Abstract
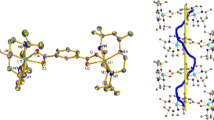
A single crystal X-ray investigation of the purple nickel (II) complex Ni[L(en)](NO3)2, produced by condensation of 1,2-diaminoethane (en) with the thiodiketone S(CH2COCH3)2 in the presence of nickel (II) nitrate (L is NH2(CH2)2N=C(CH3)CH2SCH2COCH3), shows that the metal ions are six coordinate. The new ligand L is tetradentate, with the N,N and S donor atoms arranged meridionally, and the coordination shell completed by the carbonyl oxygen atom of L and the nitrogen atoms of 1,2-diaminoethane. Metal-ligand bond distances are Ni-N: 2.08 av. (en), 2.10 and 2.05 (NH2 and N=C respectively of L); Ni-O, 2.16; Ni-S, 2.45 Å. There is considerable angular distortion from regular octahedral values of the bond angles at nickel. The crystals are monoclinic,P21/c (No. 14),a=12.433(2),b=11.709(1),c=13.150(2) Å,β=103.93(1)°,V c=1858.2(4) Å3. The structure was solved by the heavy-atom method and refined by full-matrix least squares to anR value of 0.055 for 3633 reflections.
Similar content being viewed by others
References
Sandhu, S. S., Tandon, S. S., and Singh, Harjit, (1979)Synthesis 46, 623.
Sandhu, S. S., and Hundal, M. S. (1982)Acta Cryst. B 38, 697, 842, and references therein.
Sheldrick, G. M. (1975)Shelx Program for Crystal Structure Determination, Cambridge University, England.
Tandon, S. S., and Larkworthy, L. F. (1984).J. Chem. Soc., Dalton Trans., in press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ladd, M.F.C., Larkworthy, L.F., Leonard, G.A. et al. Structure of (1,2-diaminoethane) (1-[[2-[(2-aminoethyl)imino]propyl]thio]-2-propanone)nickel(II) nitrate. Journal of Crystallographic and Spectroscopic Research 15, 19–27 (1985). https://doi.org/10.1007/BF01185713
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01185713