Abstract
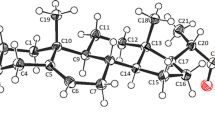
Several optically pure representatives of 2α-hydroxy-3α-[N-alkyl (aryl)] pinanoamines, potential chiral auxiliaries in organic synthesis, were obtained. In addition, a structural examination of 2α-hydroxy-3α-(N-n-butyl)-pinanoamine hydrochloride was conducted. Results of these studies confirm the structure of the amines obtained and the correctness of the proposed reaction pathway.Crystal data: C12H28NOCl,P21/c,a=13.196(1),b=8.562(1),c=14.276(1) Å,β=110.41(3)°,R=0.034 for 2046 reflections.
Similar content being viewed by others
References
Ap Simon, J. W., and LeeCollier, T. (1986)Tetrahedron 42, 5157–5254, and references cited therein.
Burak, K., and Chabudziński, Z. (1978)Pol. J. Chem. 50, 1721–1727.
Carlson, R. G. and Pierce, J. K. (1971)J. Org. Chem. 36, 2319–2324.
Duax, L., and Weeks, C. M. (1984)Atlas of Steroid Structure, Vol. 2 (IFI/Plenum Data Company).
Karle, J. and Karle, I. L. (1966)Acta Cryst. 21, 849.
Markowicz, S. W., and Bochwic, B. (1978)Pol. J. Chem. 52, 671–672.
Markowicz, S. W. (1979)Pol. J. Chem. 53, 157–161.
Markowicz, S. W. (1988) unpublished results
Markowicz, S. W., Karolak-Wojciechowska, J., and Kwiatkowski, W. (1989)J. Crystallogr. Spectrosc. Res. 19, 535–545.
Sheldrick, G. M. (1976)System of Programmes for Crystal Structure Determination. (Univ. of Cambridge, England).
Sheldrick, G. M. (1986)The Programme for Solution of Crystal Structures (Inst. fur Anorganische Chemie der Universitat Gottingen, Federal Rep. of Germany).
Author information
Authors and Affiliations
Additional information
Part I,J. Cryst. Spec. Res. (1989)19, 535.
Rights and permissions
About this article
Cite this article
Karolak-Wojciechowska, J., Kwiatkowski, W. & Markowicz, S.W. 2α-Hydroxy-3α-[N-alkyl(aryl)]-pinanoamines. Part II. Synthesis and the nature of some pinanoamines and crystal and molecular structure of 2α-hydroxy-3α-(N-n-butyl)-pinanoamine hydrochloride. Journal of Crystallographic and Spectroscopic Research 19, 893–903 (1989). https://doi.org/10.1007/BF01185355
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01185355