Summary
-
1.
A study has been made of the contact-catalysis reactions of hexane, heptane, and octane in presence of platinized alumina at 460° under a high pressure of hydrogen (15–20 atm).
-
2.
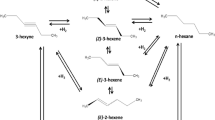
It has been shown that in all cases, under the given conditions, dehydrocyclization and isomerization of the alkanes occur, and both the alkanes themselves, and also benzene homologs formed as a result of catalytic dehydrocyclization, undergo methylation and hydrogenolysis.
-
3.
It has been suggested that the methylene breakdown of alkanes and cycloalkanes plays a part in the formation of branched paraffins and higher benzene homologs.
-
4.
A new concept has been advanced relating to the contact-catalysis isomerization reactions of alkanes at high temperatures in presence of hydrogen at high presure.
Similar content being viewed by others
Literature cited
N. I. Shuikin, N. G. Berdnikova, and S. S. Novikov, Bull. Acad. Sci. USSR, Div. Chem. Sci., No. 2, 271 (1953).
B. I. Moldavsky, G. L. Kanusher, and M. V. Kobylskaya, J. Gen. Chem., 7, 167 (1937).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shuikin, N.I., Berdnikova, N.G. & Novikov, S.S. High-temperature contact-catalysis reactions of normal alkanes in presense of hydrogen at high pressure. Russ Chem Bull 2, 779–786 (1953). https://doi.org/10.1007/BF01178857
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01178857