Summary
-
1.
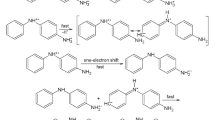
A study was made of the mechanism of the action of reversible oxidation-reduction systems consisting of an organic hydroperoxide, a dienol, and an iron or copper salt.
-
2.
It was shown that it is possible to use these systems for the initiation of polymerization at low temperatures.
Similar content being viewed by others
Literature cited
V. A. Kozlov and B. V. Ptitsyn, J. Anal. Chem. 4, 35 (1949).
H. W. Laundrie, F. Cann, Ind, Eng. Chem. 41, 8, 1568 (1949).
C. Marvel, R. Deanin, J. Pol. Sci. 3, 1, 123 (1948).
B. A. Dolgoplosk and E. A. Kropacheva, J. Gen. Chem. 26, 11, 2890 (1956.
D. M. Mikhlin, Peroxides and Peroxidases, 1948, Acad. Sci. USSR Press.
M. A. Bodin, Fact. Lab. 8, 32 (1939).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Belonovskaya, G.P., Dolgoplosk, B.A. & Tinyakova, E.I. Oxidation-reduction systems for the initiation of free-radical processes Communication 1. Reversible systems in which organic hydroperoxides, dienols, and salts of metals of variable valence take part. Russ Chem Bull 5, 1521–1528 (1956). https://doi.org/10.1007/BF01177534
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01177534