Summary
-
1.
The systems InCl3-Rb4 [Fe (CN)6]-H2O and InCl3-Cs4 [Fe (CN)6]-H2O were studied by the methods of solubility, electrical conductivity, and potentiometric and light-extinction measurements.
-
2.
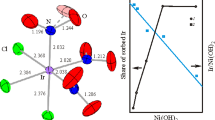
In the system InCl3-Rb4 [Fe (CN)6]-H2O indium ions are found to react with formation of solid solutions and a mixed ferrocyanide with the composition Rbln [Fe (CN)6].
-
3.
The mixed ferrocyanide Csln [Fe (CN)6] is formed in the system InCl3-Cs4 [Fe (CN)6]-H2O; a solid solution is also formed and (with suitable ratios of components) a second mixed ferrocyanide Cs7In3 [Fe (CN)6]4.
-
4.
Indium ferrocyanides were analyzed by chemical, crystallographic and x-ray methods.
Similar content being viewed by others
Literature cited
E. N. Deichman and I. V. Tananaev, Chemistry of Rare Elements, 1955, p. 2, 37.
A. G. Elitsur, Factory Lab. 12, 794 (1946).
J. Mitchell and D. Smith, Aquametry (Foreign Lit. Press, Moscow, 1952), p, 27.
I. V. Tananaev and M. I. Levina, J. Appl. Chem. 17, 356 (1944).
V. G. Kuznetsov, Report of Institute of General and Inorganic Chemistry, 1956.
E. N. Deichman and I. V. Tananaev, Volumetric and Colorimetric Methods of Determination of Indium, Report of Institute of General and Inorganic Chemistry 1, 1.956.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Deichman, E.N. Mixed ferrocyanides of indium with rubidium and cesium. Russ Chem Bull 6, 1044–1051 (1957). https://doi.org/10.1007/BF01169380
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01169380